Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Hiv-il Ritorno Dei Morti Viventi
- Creatore Discussione martino72
- Data di inizio
Più opzioni
Chi ha risposto?martino72
abighedurenosiresone
- Registrato
- 15/9/05
- Messaggi
- 3.061
- Punti reazioni
- 142
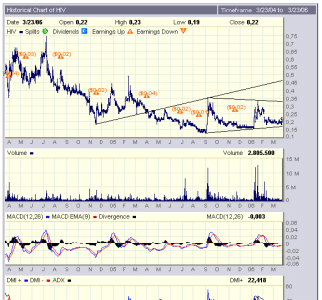
volumi buoni... 
+20% circa...
a pochi giorni dall'approvazione dell'fda cinese (27 o 28 marzo).....
se tanto mi da tanto.....


il nome del 3d fa schifo, mi sono pentito 2 secondi dopo....ma sono un cultore del genere e l'idea mi piaceva........


buona fortuna a tutti....ora credo che veramente siamo agli sgoccioli....o si va o si sta alla grande.........





+20% circa...

a pochi giorni dall'approvazione dell'fda cinese (27 o 28 marzo).....
se tanto mi da tanto.....
il nome del 3d fa schifo, mi sono pentito 2 secondi dopo....ma sono un cultore del genere e l'idea mi piaceva........
buona fortuna a tutti....ora credo che veramente siamo agli sgoccioli....o si va o si sta alla grande.........





ciao Martì.......
buona serata a tutti................


come dicevo qlke giorno fa, quello che conta sono i VOLUMI......





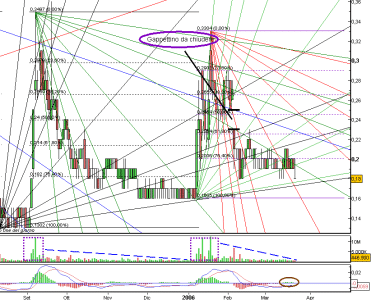
Abbiamo toccato i 0,23, volumi molto interessanti e sembra che siamo al terzo forte movim. UP da SETT.05. Quello che bolle in pentola lo sappiamo ormi tutti molto bene, il fatto che sia entrato quell'hedge f. con 35 milioni di pezzi a fine 2005 la dice anche lunga su molte cose.......


Il target di 0,45 è tutt'altro che una semplice ipotesi.......



Gian
martino72 ha scritto:giancarlino dove sei in questi momenti di (rari su hiv) di gloria???????
oggi ero in ferieeeeeeee
buona serata a tutti................
come dicevo qlke giorno fa, quello che conta sono i VOLUMI......
Abbiamo toccato i 0,23, volumi molto interessanti e sembra che siamo al terzo forte movim. UP da SETT.05. Quello che bolle in pentola lo sappiamo ormi tutti molto bene, il fatto che sia entrato quell'hedge f. con 35 milioni di pezzi a fine 2005 la dice anche lunga su molte cose.......
Il target di 0,45 è tutt'altro che una semplice ipotesi.......


Gian
Allegati
martino72
abighedurenosiresone
- Registrato
- 15/9/05
- Messaggi
- 3.061
- Punti reazioni
- 142
quando parti sei inarrestabbbbbbileeeegiumi69 ha scritto:ciò che in fondo interessa a chi investe........





Gian




matteobarison
Nuovo Utente
- Registrato
- 15/10/03
- Messaggi
- 864
- Punti reazioni
- 18
ragazzi scusate forse anche la domanda un po stupida ma siete sicuri che il 27 o 28 Marzo esce la notizia sulla cina e se rimane nell'amex oppure no??
martino72
abighedurenosiresone
- Registrato
- 15/9/05
- Messaggi
- 3.061
- Punti reazioni
- 142
matteobarison ha scritto:ragazzi scusate forse anche la domanda un po stupida ma siete sicuri che il 27 o 28 Marzo esce la notizia sulla cina e se rimane nell'amex oppure no??
secondo i "MEGLIO INFORMATI" dovrebbe essere così....
"Looks like they are using 45 working days (35 + 10) or 70+/- days starting from Jan. 16. So March 26-March 28 is the date for the news."
crypta
NIHIL CREDENDUM,...
- Registrato
- 31/8/05
- Messaggi
- 3.349
- Punti reazioni
- 567
AMEX Agrees Not to Delist Calypte's Common Stock
2006/03/24 08:31:02
LAKE OSWEGO, Ore., March 24, 2006 /PRNewswire-FirstCall via COMTEX/ -- Calypte Biomedical Corporation (Amex: HIV) announced today that it has received a letter from the American Stock Exchange (the "Amex") regarding the results of the March 13, 2006 hearing conducted before a listing qualifications panel of the Amex Committee on Securities (the "Panel").
The Panel unanimously agreed that the Amex should not move to delist Calypte's common stock at this time and that it would allow the Company until July 31, 2006 to regain compliance with the Amex's continued listing standards. By that date, Calypte must demonstrate that it has regained compliance with the Amex's continued listing standards. If Calypte cannot demonstrate at that time that it is in compliance with those standards, the Panel agreed that the Amex should immediately move to delist Calypte's common stock.
The Company intends to focus on executing its business plan during this time and building stockholder value. However, there can be no assurance that the Company will be able to meet the continued listing standards required to retain the listing of its common stock on the Amex.
Mr. Roger Gale, Chairman and Chief Executive Officer said, "We are grateful that the Amex Panel understood the importance of the coming months and has allowed us additional time to regain compliance with the Amex's listing standards. We believe that the events in the coming quarter will allow us to do so."
2006/03/24 08:31:02
LAKE OSWEGO, Ore., March 24, 2006 /PRNewswire-FirstCall via COMTEX/ -- Calypte Biomedical Corporation (Amex: HIV) announced today that it has received a letter from the American Stock Exchange (the "Amex") regarding the results of the March 13, 2006 hearing conducted before a listing qualifications panel of the Amex Committee on Securities (the "Panel").
The Panel unanimously agreed that the Amex should not move to delist Calypte's common stock at this time and that it would allow the Company until July 31, 2006 to regain compliance with the Amex's continued listing standards. By that date, Calypte must demonstrate that it has regained compliance with the Amex's continued listing standards. If Calypte cannot demonstrate at that time that it is in compliance with those standards, the Panel agreed that the Amex should immediately move to delist Calypte's common stock.
The Company intends to focus on executing its business plan during this time and building stockholder value. However, there can be no assurance that the Company will be able to meet the continued listing standards required to retain the listing of its common stock on the Amex.
Mr. Roger Gale, Chairman and Chief Executive Officer said, "We are grateful that the Amex Panel understood the importance of the coming months and has allowed us additional time to regain compliance with the Amex's listing standards. We believe that the events in the coming quarter will allow us to do so."
matteobarison
Nuovo Utente
- Registrato
- 15/10/03
- Messaggi
- 864
- Punti reazioni
- 18
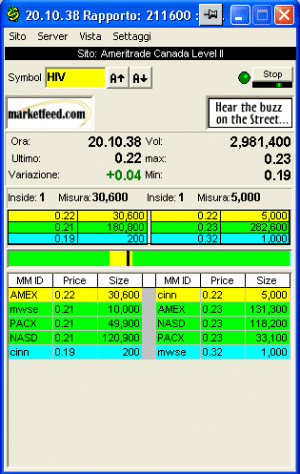
lo spero io le ho riprese a 0,27 !!!!!!!!!!!!
superbaffone
1000k
- Registrato
- 23/7/03
- Messaggi
- 11.440
- Punti reazioni
- 1.120
Calypte's Aware(TM) Rapid HIV-1/2 Tests Receive Approval in the Republic of Kenya
PR Newswire - March 28, 2006 08:30
LAKE OSWEGO, Ore., March 28, 2006 /PRNewswire-FirstCall via COMTEX/ -- Calypte Biomedical Corporation (Amex: HIV) announced today the Kenyan National Public Health Laboratory Services (NPHLS) has issued notification of registration and approval for the Calypte Aware(TM) HIV-1/2 BSP (blood) and Aware(TM) HIV-1/2 OMT (oral fluid) rapid tests in the Republic of Kenya.
The registration and approval follows a formal evaluation of the products by The National AIDS Control Programme (NASCOP) of the Kenyan Ministry of Health. NASCOP recently completed a formal evaluation of the tests and found them to "meet the requirements for sensitivity and specificity for HIV test kits in Kenya." The kits are "certified for testing [sale] in Kenya."
E. M. Kiragu, Director of UltraLab and Allied Services, our distributor for Kenya said, "We are delighted that the Aware(TM) HIV-1/2 assays for blood and for oral fluid have now been registered here in Kenya and are available for sale. The oral fluid assay particularly has generated tremendous interest."
Roger Gale, Chairman and Chief Executive Officer stated, "With this announcement Calypte continues to expand the breadth of countries where we can market our rapid HIV-1/2 diagnostic tests. Kenya is an important milestone as a key country in the fight against AIDS in sub-Saharan Africa. Further, the positive clinical result from Kenya adds an important base to our worldwide clinical results, building to our planned upcoming application to the USAID Waiver list. In addition to enabling these products to compete for Kenyan government tenders expected to exceed over 4 million tests when announced later this year, our distributor has already contacted several NGOs who can buy these products directly now that they are registered. I look forward to updating investors on all of our progress in the past months in the coming days. We expect to file our 10KSB around the end of the month and will announce a conference call to discuss those results at the same time."
PR Newswire - March 28, 2006 08:30
LAKE OSWEGO, Ore., March 28, 2006 /PRNewswire-FirstCall via COMTEX/ -- Calypte Biomedical Corporation (Amex: HIV) announced today the Kenyan National Public Health Laboratory Services (NPHLS) has issued notification of registration and approval for the Calypte Aware(TM) HIV-1/2 BSP (blood) and Aware(TM) HIV-1/2 OMT (oral fluid) rapid tests in the Republic of Kenya.
The registration and approval follows a formal evaluation of the products by The National AIDS Control Programme (NASCOP) of the Kenyan Ministry of Health. NASCOP recently completed a formal evaluation of the tests and found them to "meet the requirements for sensitivity and specificity for HIV test kits in Kenya." The kits are "certified for testing [sale] in Kenya."
E. M. Kiragu, Director of UltraLab and Allied Services, our distributor for Kenya said, "We are delighted that the Aware(TM) HIV-1/2 assays for blood and for oral fluid have now been registered here in Kenya and are available for sale. The oral fluid assay particularly has generated tremendous interest."
Roger Gale, Chairman and Chief Executive Officer stated, "With this announcement Calypte continues to expand the breadth of countries where we can market our rapid HIV-1/2 diagnostic tests. Kenya is an important milestone as a key country in the fight against AIDS in sub-Saharan Africa. Further, the positive clinical result from Kenya adds an important base to our worldwide clinical results, building to our planned upcoming application to the USAID Waiver list. In addition to enabling these products to compete for Kenyan government tenders expected to exceed over 4 million tests when announced later this year, our distributor has already contacted several NGOs who can buy these products directly now that they are registered. I look forward to updating investors on all of our progress in the past months in the coming days. We expect to file our 10KSB around the end of the month and will announce a conference call to discuss those results at the same time."