M
Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Htgm
- Creatore Discussione coffeman
- Data di inizio
Più opzioni
Chi ha risposto?
M
Membro cancellato 234088
Guest
Partita
mastrodaniel
Nuovo Utente
- Registrato
- 21/9/05
- Messaggi
- 6.012
- Punti reazioni
- 247

 u
uThe Miracle
The game never ends
- Registrato
- 17/9/00
- Messaggi
- 13.406
- Punti reazioni
- 966
mastrodaniel
Nuovo Utente
- Registrato
- 21/9/05
- Messaggi
- 6.012
- Punti reazioni
- 247
Logicamente già out un gain non si butta
coffeman
Nuovo Utente
- Registrato
- 14/6/04
- Messaggi
- 29.916
- Punti reazioni
- 1.197
beppofinanza
Nuovo Utente
- Registrato
- 7/1/21
- Messaggi
- 878
- Punti reazioni
- 49
Logicamente già out un gain non si butta
grande mastro!!.. uscito anche io 0.8 non mi ero accorto della sparata a 0.99.. ma va bene cosi
The Miracle
The game never ends
- Registrato
- 17/9/00
- Messaggi
- 13.406
- Punti reazioni
- 966
Uscito ad 85c.. un po' troppo presto, ora è 1$.
Solita vecchia regola: se guadagno non mi lagno. ( A patto che non siano briciole ).
Solita vecchia regola: se guadagno non mi lagno. ( A patto che non siano briciole ).
M
Membro cancellato 234088
Guest
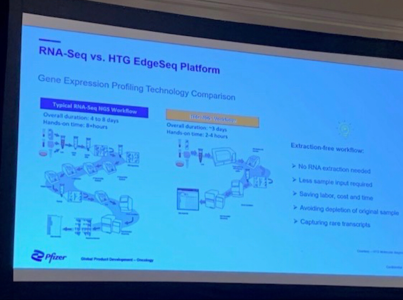
Ahhhhhh, mamma mia che occasione questa. La collaborazione con Pfizer sarebbe stato un bel campanello per andare long a lungo.
Peccato non abbiano rilasciato molte info sulla presentazione fatta, la schermata che avevo postato l'ho trovata per vie traverse chiedendo un approfondimento su un commento su Stockwits e per fortuna quella buon'anima a cui ho chiesto info ha deciso di postarla, altrimenti si faticava a trovare riscontri.
Peccato non abbiano rilasciato molte info sulla presentazione fatta, la schermata che avevo postato l'ho trovata per vie traverse chiedendo un approfondimento su un commento su Stockwits e per fortuna quella buon'anima a cui ho chiesto info ha deciso di postarla, altrimenti si faticava a trovare riscontri.
M
Membro cancellato 234088
Guest
Oggi sono rientrato, in after c’è un bel movimento
serenella6
Nuovo Utente
- Registrato
- 18/5/09
- Messaggi
- 10.636
- Punti reazioni
- 504
Oggi sono rientrato, in after c’è un bel movimento
Se vuoi vedere i bei movimenti, guarda , il titolo che avevo segnalato a coffe qualche settimana fa
HKD..Cius..Cius
getulio
Nuovo Utente
- Registrato
- 2/4/20
- Messaggi
- 103
- Punti reazioni
- 38
Se vuoi vedere i bei movimenti, guarda , il titolo che avevo segnalato a coffe qualche settimana faOggi da 46$ a 287$..una montagna di soldi..nei trading garage New Yorkesi
HKD..Cius..Cius
 :sbonk:
:sbonk:
M
Membro cancellato 234088
Guest
TUCSON, Ariz., Sept. 20, 2022 (GLOBE NEWSWIRE) -- HTG Molecular Diagnostics, Inc. (NASDAQ:HTGM) (HTG), a life science company advancing precision medicine through its innovative transcriptome-wide profiling technology, announced that the Fundación Instituto Valenciano de Oncología, located in Valencia, Spain (IVO), with its partners, the Fundación Pública Andaluza Progreso y Salud (FPS) and the Centro de Investigación Biomédica en Red (CIBER), have developed a laboratory-developed test (LDT) (the MPD Test) for the assessment of breast cancer recurrence based on their use of the HTG EdgeSeq™ technology.
This innovative test for patients with HR+/HER2- early-stage breast cancer was developed by IVO and its partners to identify patients with an increased risk of developing distant metastasis or relapse who may benefit from adjuvant chemotherapy. IVO is among the top 50 cancer hospitals in the world; FPS is a public entity mainly dedicated to research and information technology; and CIBER is a public research consortium devoted to advancing research in biomedicine and health sciences.
This highly qualified and experienced team of researchers has partnered to develop and validate the MPD Test using the HTG EdgeSeq technology, which typically requires only one single 5 μm-thin FFPE tissue biopsy section, coupled with the sensitivity and dynamic range of next-generation sequencing (NGS)-based detection, to generate results in as little as 3 days.
"We are thrilled that three of Spain's most renowned cancer research organizations have chosen to leverage HTG's innovative technology to improve precision medicine for breast cancer patients," said Byron Lawson, Senior Vice President and Chief Commercial Officer of HTG. "The MPD Test is a great example of how the HTG EdgeSeq technology can be used not only for molecular profiling and analysis, but also as a predictive tool for advancing precision medicine."
"Among the challenges of traditional genomic testing in early-stage HR+/HER2- breast cancer is the wide variation in risk assessment," said Dr. José Antonio López-Guerrero, Head of Molecular Biology Laboratory at IVO. "Discordant results between two different genomic assays can have a significant clinical impact on patients, determining, for example, whether or not a patient chooses to undergo chemotherapy. Leveraging HTG's EdgeSeq technology, we believe that the MPD Test represents a cost-effective option to shorten the time to result, reduce the risk of potential bias and improve the overall risk profile using only a small sample from a tissue biopsy."
The MPD Test is available as an LDT provided by IVO and its partners.
This innovative test for patients with HR+/HER2- early-stage breast cancer was developed by IVO and its partners to identify patients with an increased risk of developing distant metastasis or relapse who may benefit from adjuvant chemotherapy. IVO is among the top 50 cancer hospitals in the world; FPS is a public entity mainly dedicated to research and information technology; and CIBER is a public research consortium devoted to advancing research in biomedicine and health sciences.
This highly qualified and experienced team of researchers has partnered to develop and validate the MPD Test using the HTG EdgeSeq technology, which typically requires only one single 5 μm-thin FFPE tissue biopsy section, coupled with the sensitivity and dynamic range of next-generation sequencing (NGS)-based detection, to generate results in as little as 3 days.
"We are thrilled that three of Spain's most renowned cancer research organizations have chosen to leverage HTG's innovative technology to improve precision medicine for breast cancer patients," said Byron Lawson, Senior Vice President and Chief Commercial Officer of HTG. "The MPD Test is a great example of how the HTG EdgeSeq technology can be used not only for molecular profiling and analysis, but also as a predictive tool for advancing precision medicine."
"Among the challenges of traditional genomic testing in early-stage HR+/HER2- breast cancer is the wide variation in risk assessment," said Dr. José Antonio López-Guerrero, Head of Molecular Biology Laboratory at IVO. "Discordant results between two different genomic assays can have a significant clinical impact on patients, determining, for example, whether or not a patient chooses to undergo chemotherapy. Leveraging HTG's EdgeSeq technology, we believe that the MPD Test represents a cost-effective option to shorten the time to result, reduce the risk of potential bias and improve the overall risk profile using only a small sample from a tissue biopsy."
The MPD Test is available as an LDT provided by IVO and its partners.
Salva_Tori
Nuovo Utente
- Registrato
- 12/6/07
- Messaggi
- 10.870
- Punti reazioni
- 282
TUCSON, Ariz., Sept. 20, 2022 (GLOBE NEWSWIRE) -- HTG Molecular Diagnostics, Inc. (NASDAQ:HTGM) (HTG), a life science company advancing precision medicine through its innovative transcriptome-wide profiling technology, announced that the Fundación Instituto Valenciano de Oncología, located in Valencia, Spain (IVO), with its partners, the Fundación Pública Andaluza Progreso y Salud (FPS) and the Centro de Investigación Biomédica en Red (CIBER), have developed a laboratory-developed test (LDT) (the MPD Test) for the assessment of breast cancer recurrence based on their use of the HTG EdgeSeq™ technology.
This innovative test for patients with HR+/HER2- early-stage breast cancer was developed by IVO and its partners to identify patients with an increased risk of developing distant metastasis or relapse who may benefit from adjuvant chemotherapy. IVO is among the top 50 cancer hospitals in the world; FPS is a public entity mainly dedicated to research and information technology; and CIBER is a public research consortium devoted to advancing research in biomedicine and health sciences.
This highly qualified and experienced team of researchers has partnered to develop and validate the MPD Test using the HTG EdgeSeq technology, which typically requires only one single 5 μm-thin FFPE tissue biopsy section, coupled with the sensitivity and dynamic range of next-generation sequencing (NGS)-based detection, to generate results in as little as 3 days.
"We are thrilled that three of Spain's most renowned cancer research organizations have chosen to leverage HTG's innovative technology to improve precision medicine for breast cancer patients," said Byron Lawson, Senior Vice President and Chief Commercial Officer of HTG. "The MPD Test is a great example of how the HTG EdgeSeq technology can be used not only for molecular profiling and analysis, but also as a predictive tool for advancing precision medicine."
"Among the challenges of traditional genomic testing in early-stage HR+/HER2- breast cancer is the wide variation in risk assessment," said Dr. José Antonio López-Guerrero, Head of Molecular Biology Laboratory at IVO. "Discordant results between two different genomic assays can have a significant clinical impact on patients, determining, for example, whether or not a patient chooses to undergo chemotherapy. Leveraging HTG's EdgeSeq technology, we believe that the MPD Test represents a cost-effective option to shorten the time to result, reduce the risk of potential bias and improve the overall risk profile using only a small sample from a tissue biopsy."
The MPD Test is available as an LDT provided by IVO and its partners.
notizie positive ma il titolo reagisce all' opposto

M
Membro cancellato 234088
Guest
Intanto vediamo oggi, l'after di ieri era piuttosto farlocco visti i volumi pressoché nulli.
M
Membro cancellato 234088
Guest
M
Membro cancellato 234088
Guest
Volume al momento: 105 
M
Membro cancellato 234088
Guest
Occhio qui
M
Membro cancellato 234088
Guest
On November 18, 2022, HTG Molecular Diagnostics, Inc. (the “Company”) received notice (the “Notice”) from The Nasdaq Stock Market LLC (“Nasdaq”) that, as a result of the Company’s stockholders’ equity falling below $2.5 million, as reported in the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2022, the Company does not satisfy one of The Nasdaq Capital Market continued listing requirement set forth in Nasdaq Stock Market Rule 5550(b) (the “Rule”).
Pursuant to the Notice and Nasdaq rules, the Company has 45 calendar days, or until January 2, 2023, to submit a plan to regain compliance with the Rule. If the plan is accepted, Nasdaq may grant an extension of up to 180 calendar days from the date of the Notice for the Company to provide evidence of compliance. There can be no assurance that the Company will be able to regain compliance with the Rule.
Pursuant to the Notice and Nasdaq rules, the Company has 45 calendar days, or until January 2, 2023, to submit a plan to regain compliance with the Rule. If the plan is accepted, Nasdaq may grant an extension of up to 180 calendar days from the date of the Notice for the Company to provide evidence of compliance. There can be no assurance that the Company will be able to regain compliance with the Rule.