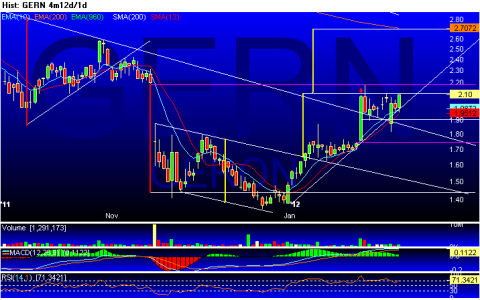
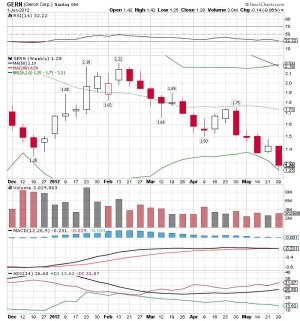
Geron Corporation Reports Fourth Quarter and Annual 2011 Financial Results - Yahoo! Finance
MENLO PARK, Calif., March 7, 2012 - Geron Corporation (Nasdaq: GERN - News) today reported financial results for the fourth quarter and year ended December 31, 2011.
Fourth Quarter 2011 Results
Net loss for the fourth quarter of 2011 was $31.9 million, or $0.25 per share, compared to $59.4 million, or $0.59 per share, for the comparable 2010 period. Net loss for the fourth quarter of 2011 included restructuring charges of $5.4 million related to discontinuation of the company`s stem cell programs and non-cash debt extinguishment charges of $1.7 million resulting from the repayment of the loan to the California Institute for Regenerative Medicine. Net loss for the fourth quarter of 2010 included a charge of $35.0 million in connection with an in-licensing transaction with Angiochem, Inc. The company ended the year with $154.2 million in cash and investments.
In the fourth quarter of 2011, the company had revenues of $251,000, compared to $1.1 million for the comparable 2010 period. Revenues in both periods primarily reflected license fees and royalties. Revenues for the fourth quarter of 2010 also included funding under a collaborative agreement.
Interest and other income for the fourth quarter of 2011 was $204,000, compared to $1.4 million for the comparable 2010 period. In November 2010, the company received $1.2 million in total grants under the Qualifying Therapeutic Discovery Project (QTDP) program.
In the fourth quarter of 2011, the company had operating expenses of $30.7 million, including restructuring charges of $5.4 million, compared to operating expenses of $60.7 million for the comparable 2010 period, which included a charge of $35.0 million in connection with the in-license from Angiochem. Research and development expenses for the fourth quarter of 2011 were $19.7 million, compared to $21.0 million for the comparable 2010 period. The decrease in research and development expenses was due primarily to reduced clinical drug product purchases and manufacturing costs and lower scientific supply expenses as a result of discontinuation of the stem cell programs. General and administrative expenses for the fourth quarter of 2011 were $5.5 million, compared to $4.7 million for the comparable 2010 period. The increase was due primarily to higher personnel-related expenses in connection with management transitions.
Non-cash operating expenses for the fourth quarter of 2011 were approximately $6.0 million, which primarily included stock-based compensation, write-downs of excess lab equipment related to the stem cell programs and expense for stock issued for services. Non-cash operating expenses for the fourth quarter of 2010 were approximately $38.4 million, which primarily included expense for the in-license from Angiochem, stock-based compensation and expense for stock issued for services.
Year End 2011 Results
For 2011, net loss was $96.9 million, or $0.78 per share, which included restructuring charges of $5.4 million and non-cash debt extinguishment charges of $1.7 million, compared to $111.4 million, or $1.14 per share, for 2010, which included a charge of $35.0 million in connection with the in-license from Angiochem.
Revenues for 2011 and 2010 were $2.4 million and $3.6 million, respectively, reflecting funding under a collaborative agreement and license fees and royalties.
For 2011, interest and other income was $1.0 million, compared to $2.0 million for 2010, which included $1.2 million in total grants under the QTDP program.
For 2011, the company had operating expenses of $98.6 million, including restructuring charges of $5.4 million, compared to $114.7 million for 2010, which included a charge of $35.0 million in connection with the in-license from Angiochem. Research and development expenses for 2011 were $69.3 million, compared to $61.7 million for 2010. Overall, research and development expenses increased in 2011 as a net result of higher clinical drug product purchases and clinical trial costs, partially offset by reduced scientific supply costs as a result of discontinuation of the stem cell programs and lower non-cash compensation expense for stock-based awards. General and administrative expenses for 2011 were $23.8 million, compared to $18.0 million for 2010. The increase was primarily the result of higher non-cash compensation expense and personnel-related costs in connection with management transitions during the year and increased legal and consulting costs.
Non-cash operating expenses for 2011 were approximately $25.2 million, which primarily included stock-based compensation, depreciation, write-downs of excess lab equipment related to the stem cell programs and expense for stock issued for services. Non-cash operating expenses for 2010 were approximately $59.2 million, which primarily included expense for the in-license from Angiochem, stock-based compensation, depreciation and expense for stock issued for services.






 http://www.finanzaonline.com/forum/...-corporation-nasdaq-gern-11.html#post31165303
http://www.finanzaonline.com/forum/...-corporation-nasdaq-gern-11.html#post31165303