Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
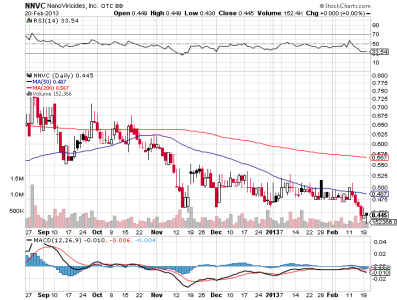
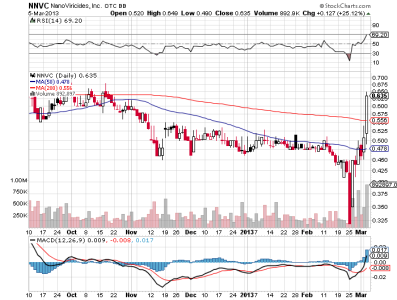
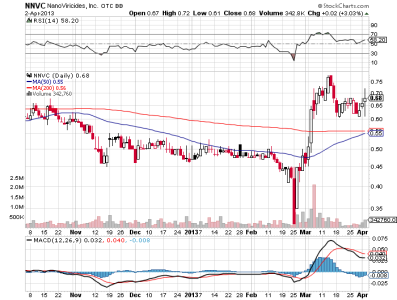
nnvc
- Creatore Discussione axelvento
- Data di inizio
-
 Ecco la 67° Edizione del settimanale "Le opportunità di Borsa" dedicato ai consulenti finanziari ed esperti di borsa.
Ecco la 67° Edizione del settimanale "Le opportunità di Borsa" dedicato ai consulenti finanziari ed esperti di borsa.
 Nell’ultima settimana borsistica, i principali indici globali hanno messo a segno performance positive. In assenza di dati macro di rilievo, gli operatori si sono focalizzati sugli utili societari e sulle banche centrali. La stagione delle trimestrali è infatti entrata nel vivo in Europa e a Piazza Affari con oltre la metà dei 40 titoli che compongono il Ftse Mib ad alzare il velo sui conti. Per quanto riguarda le banche centrali, la Reserve Bank of Australia ha lasciato i tassi di interesse invariati, come previsto. Anche la Bank of England ha lasciato fermi i tassi, con due voti a favore di un taglio immediato sui nove totali. La Riksbank svedese ha invece tagliato i tassi per la prima volta in otto anni, riducendo il costo del denaro di 25 punti base al 3,75%, evidenziando la divergenza dell’Europa dalla linea dura della Fed. Per continuare a leggere visita il link
Nell’ultima settimana borsistica, i principali indici globali hanno messo a segno performance positive. In assenza di dati macro di rilievo, gli operatori si sono focalizzati sugli utili societari e sulle banche centrali. La stagione delle trimestrali è infatti entrata nel vivo in Europa e a Piazza Affari con oltre la metà dei 40 titoli che compongono il Ftse Mib ad alzare il velo sui conti. Per quanto riguarda le banche centrali, la Reserve Bank of Australia ha lasciato i tassi di interesse invariati, come previsto. Anche la Bank of England ha lasciato fermi i tassi, con due voti a favore di un taglio immediato sui nove totali. La Riksbank svedese ha invece tagliato i tassi per la prima volta in otto anni, riducendo il costo del denaro di 25 punti base al 3,75%, evidenziando la divergenza dell’Europa dalla linea dura della Fed. Per continuare a leggere visita il link
Più opzioni
Chi ha risposto?playars
lotto players
- Registrato
- 11/4/08
- Messaggi
- 8.306
- Punti reazioni
- 174
playars
lotto players
- Registrato
- 11/4/08
- Messaggi
- 8.306
- Punti reazioni
- 174
The Company has previously announced that Seaside 88, LP, a Florida limited partnership (“Seaside”), exercised their option to purchase an additional $2.5M of the Company’s Series B Convertible Preferred Stock. This amounted to net proceeds of approximately $2.295M to the Company after expensing commissions and legal fees. Seaside has also agreed to purchase an additional $2.5M of the Company’s Series B Convertible Preferred Stock within 90 days of the closing of the first follow on sale of the Company’s Series B Preferred Stock. The Series B Preferred Stock is convertible into a number of shares of the Company’s common stock every two weeks. The converted shares are estimated to represent less than five percent of the 10-day trading volume of the Company’s stock (OTC BB: NNVC.OB), based upon current data.
The Company has no long term debt. The Company estimates that it currently has sufficient cash in hand to achieve the budgeted objectives for over 18 months of operations.
The Company has reported satisfactory progress in its drug development objectives during this period. The Company reported that its anti-Herpes drug candidates demonstrated significant efficacy in cell culture studies. The studies were performed in the laboratory of Dr. Ken Rosenthal at Northeastern Ohio Universities Colleges of Medicine and Pharmacy. Several of the anti-Herpes nanoviricides® demonstrated a dose-dependent maximal inhibition of Herpes virus infectivity in a cell culture model. Almost complete inhibition of the virus production was observed at clinically usable concentrations. These studies employed the H129 strain of herpes simplex virus type 1 (HSV-1). H129 is an encephalitic strain that closely resembles a clinical isolate; it is known to be more virulent than classic HSV-1 laboratory strains. The Company is now working on further optimizing these drug candidates, with a view towards animal studies. The H129 strain will be used in subsequent animal testing of nanoviricides for developing a skin cream to treat oral and genital herpes outbreaks.
Subsequent to the quarterly report period, the Company recently announced that it has achieved profound levels of effectiveness with its optimized nanoviricide drug candidates against H1N1 influenza in a high infection, lethality mouse model study. The most effective FluCide candidate demonstrated a fifteen-fold (15X) greater lung viral load reduction as compared to Tamiflu, and a thirty-fold (30X) greater viral load reduction as compared to untreated animals. Tamiflu demonstrated a viral load reduction of only twofold (2X) compared to the untreated animals in this high infection, lethality study. This profound decrease in viral load was consistent with the observed substantial increase in length of survival upon nanoviricide treatment. Animals treated with this FluCide candidate survived for as long as 18.1 days on average, compared with only 7.8 days for Tamiflu treated animals. A survival length of 21 days would be considered indefinite survival in this animal model.
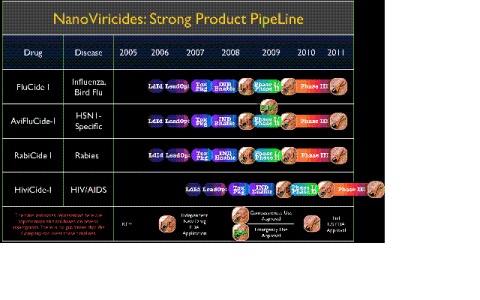
The Company currently has five commercially important drug candidates in its pipeline. These include FluCide™, HIVCide™, HerpiCide™, DengiCide™, and a broad-spectrum nanoviricide eye drop formulation against viral infections of the eye. In addition, it continues to develop its novel ADIF™ (“Accurate Drug In Field”™) technologies which promise a way to attack novel viruses, whether man-made (bioterrorism) or natural (such as SARS), before they cause a pandemic. The Company is also developing broad-spectrum therapeutics against a number of different hemorrhagic viruses such as Ebola/Marburg, and viruses causing Neglected Tropical Diseases such as Rabies.
“Now that we have a strong and wide pipeline, we plan to focus on taking our drug candidates further forward into the regulatory process,” said Eugene Seymour, MD, MPH, CEO of the Company, adding, “We have the necessary cash in hand for our immediate goals.”
About NanoViricides:
NanoViricides, Inc. (Welcome to NanoViricides, Inc.) is a development stage company that is creating special purpose nanomaterials for antiviral therapy. The Company's novel nanoviricide® class of drug candidates are designed to specifically attack enveloped virus particles and to dismantle them. The Company is developing drugs against a number of viral diseases including H1N1 swine flu, H5N1 bird flu, seasonal Influenza, HIV, oral and genital Herpes, viral diseases of the eye including EKC and herpes keratitis, Hepatitis C, Rabies, Dengue fever, and Ebola virus, among others.
playars
lotto players
- Registrato
- 11/4/08
- Messaggi
- 8.306
- Punti reazioni
- 174
playars
lotto players
- Registrato
- 11/4/08
- Messaggi
- 8.306
- Punti reazioni
- 174
.....
Allegati
playars
lotto players
- Registrato
- 11/4/08
- Messaggi
- 8.306
- Punti reazioni
- 174
Hey all my Ihub friends just sent Dr. Seymour a little Holiday note and this is his reply !!!!!!
On Dec 23, 2010, at 11:21 AM
Dear Dr. Seymour I just wanted to take a moment to wish you and your family a very Merry Christmas and to thank you for all the time and effort you put into NNVC each and every day of your life. NNVC is going to be a life changer for us all and it keeps me soooooooooooooo EXCITED !!!!!!!!!!! Something else that has me VERY excited is that NNVC has been invited to present at the Dengue Research meeting sponsored by the National Institutes Of Health. I would love to be there because I'm feeling something HUGE could be announced since Dr. Eva Harris of UC Berkeley and Dr. Harold Margolis Chief Of Dengue Branch Of CDC are attending !!!!!!!!! Well February is just around the corner and I CAN'T WAIT !!!!!!!
Take care,
Sincerely,
Patricia
Certainly OK to share
All of this is public information
Dear Patricia,
Thanks for the kind wishes.
I'm very excited about the prospects for the Company in 2011.
We've gained a reputation in the scientific community for doing innovative work in viral therapeutics.
Everything we're now doing is focused on document preparation for the FDA filings. That includes lots of laboratory and animal data on our various lead candidates.
Some have complained that we're moving too slowly.
I think that for a 5 year old company to have 9 drugs in the pipeline with 5 lead candidates for sequencing into the FDA and having done this for around $17M +/- is nothing short of amazing. And each of those candidates addresses a $1B+ market! We're actively seeking analytic and pharmaceutical chemists. I'm amazed that so many of those people feel more comfortable working for a large company where layoffs happen all the time rather than a rapid growth entrepreneurial company with some amazing technology. More people would enable us to move faster although we're still constrained by the available time in the University and government animal labs!
That being said, 2011 should be a terrific year
Sincerely,
EUGENE SEYMOUR, MD, MPH
Chief Executive Officer
NANOVIRICIDES, INC
Nanotechnology-based targeted anti-viral therapeutics
Welcome to NanoViricides, Inc.
eugene@nanoviricides.com
310-486-5677
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
Four days post virus infection, animals treated with three of the optimized FluCide(TM) nanoviricide drug candidates exhibited greater than 95% reduction in the number of lung lesions as compared to the infected yet untreated control animals (p-values < 0.001). In contrast,animals treated with Oseltamivir (Tamiflu(R), Roche) showed only a 50% reduction. In another significant finding, no increase in the number or size of the lung lesions was observed over the entire duration of the study in the FluCide(TM)-treated animals. This was not the case for the Oseltamivir-treated animals. This demonstrated that treatment with FluCide drug candidates provided clear and strong protection against lung damage caused by the severe influenza infection.
The studies were conducted by Dr. Krishna Menon, PhD, VMD, MRCS, at KARD Scientific, MA. One million virus particles of Influenza A Strain A/WS/33 (H1N1) were aspirated directly into the lungs of mice. The same quantity of virus infection was repeated at 22 hrs. This influenza model was designed to be uniformly fatal in 100% of the infected, untreated animals within 5 days after infection. Treatment with the FluCide candidates and Oseltamivir commenced 24 hours after the first viral infection. The duration of study was 21 days.
The Company had previously reported that the same three optimized FluCide(TM) nanoviricide drug candidates achieved significantly increased survival (20.2 to 22.2 days) as compared to animals treated with Oseltamivir (only 8.3 day survival). The lung histology (microscopic tissue examination) data show that the observed increase in survival was accompanied by a dramatic reduction in virus-induced lung inflammation and necrosis.
"Protection of the FluCide-treated animals against the development of influenza-specific lung inflammation and necrosis, together with the markedly extended survival clearly demonstrate the therapeutic potential of these FluCide nanoviricide drug candidates," said Dr. Krishna Menon, KARD Scientific, adding, "Importantly, there were no drug-related side effects observed in the FluCide-treated animals."
Pulmonary viral load studies, another important tool for evaluation of drug effectiveness, are currently in process. The results will be reported when they become available.
"These studies have clearly demonstrated that FluCide drug candidates are significantly superior to current therapies for influenza in animal models," said Eugene Seymour, MD, MPH, CEO of the Company, adding, "Lung damage is a very important part of the pathology of influenza virus infection. Uncontrolled lung damage is associated with viral pneumonitis that, in severe cases of influenza, can lead to respiratory failure and refractory shock, the two most common causes of death. The world has seen such severe cases in the recent H1N1 pandemic, and continues to witness them with sporadic cases of H5N1 bird flu infections. We are very pleased with the finding that FluCide can protect animals against influenza viral lung damage."
The Company is presenting its work at the 2nd Annual "Encouraging Development of Therapeutics for Neglected Diseases" Conference being held in Philadelphia, PA, today, April 4th. The Company has sponsored a luncheon presentation at this Conference. Anil R. Diwan, PhD, President of the Company, will be presenting the Company's work on neglected diseases including Dengue, Rabies, Ebola, and Bird Flu (H5N1). The Company plans to include the late-breaking information discussed in this press release in the presentation.
About NanoViricides:
NanoViricides, Inc. (Welcome to NanoViricides, Inc.) is a development stage company that is creating special purpose nanomaterials for viral therapy. The Company's novel nanoviricide(R) class of drug candidates are designed to specifically attack enveloped virus particles and to dismantle them. The Company is developing drugs against a number of viral diseases including H1N1 swine flu, H5N1 bird flu, seasonal Influenza, HIV, oral and genital Herpes, viral diseases of the eye including EKC and herpes keratitis, Hepatitis C, Rabies, Dengue fever, and Ebola virus, among others.
__________________________________________________________________________
(the "Company") reports that post-infection treatment with its optimized FluCide(TM) drug candidates resulted in significant reduction in lung tissue presence of leukocytes, and in particular, that of eosinophils in a lethal influenza infection animal model.
Four days post virus infection, animals treated with three of the optimized FluCide(TM) nanoviricide drug candidates exhibited a substantial reduction in overall leukocytes in lung tissue as compared to untreated infected control. More importantly, the eosinophil count in the lung tissues of the nanoviricide treated animals was also significantly lower than untreated animals. Further, this reduced lung tissue presence of damaging immune system cells was found to persist over the entire duration of study. In contrast, animals treated with Oseltamivir (Tamiflu(R), Roche) showed reduced eosinophil and leukocyte count initially that rapidly rose to the level of untreated animals.
Eosinophil expansion occurs in response to a viral infection, and is indicative of a viral infection. Various white blood cells (leukocytes) also increase in response to a viral infection. These phenomena are part of the normal immune response. In severe influenza cases, it is thought that patients can go into a stage called "cytokine storm syndrome". This may be thought of as an all-out attack by an expanded army of white blood cells in response to an uncontrolled viral infection. In an attempt to control the viral infection, the immune system attacks the infected cells as well as nearby normal cells. This can lead to severe lung damage that may rapidly become fatal.
"The finding that FluCide drug candidates showed substantially reduced extent of white blood cells, and in particular, eosinophils, in the lung tissue, is very significant," said Randall W. Barton, PhD Immunologist, CSO of the Company, adding, "This indicates that FluCide drug candidates may be highly effective in severe cases of influenza as well as cases of bird flu, H5N1, by protecting the patient from cytokine storm phenomena."
He also observed that the reduced white blood cell and eosinophil counts were consistent with the dramatic reduction in lung lesions that the Company has recently reported.
The studies were conducted by Dr. Krishna Menon, PhD, VMD, MRCS, at KARD Scientific, MA. One million virus particles of Influenza A Strain A/WS/33 (H1N1) were aspirated directly into the lungs of mice. The same quantity of virus infection was repeated at 22 hrs. This influenza model was designed to be uniformly fatal in 100% of the infected, untreated animals within 5 days after infection. Treatment with the FluCide candidates and Oseltamivir commenced 24 hours after the first viral infection. The duration of study was 21 days. Animals were sampled at 4.5, 9.5, 13.5 and 19.5 days for histological studies.
The Company had previously reported that the same three optimized FluCide(TM) nanoviricide drug candidates achieved significantly increased survival (20.2 to 22.2 days) as compared to animals treated with Oseltamivir (only 8.3 day survival). The lung histology (microscopic tissue examination) data show that the observed increase in survival was accompanied by a dramatic reduction in virus-induced lung inflammation and necrosis.
The Company is presenting its work at the "2nd Annual NanoBusiness NYC Conference" (New York City 2011 | NanoBusiness Commercialization Association) being held in New York City, today, April 7th. Anil R. Diwan, PhD, President of the Company, will be presenting the Company's work as a pioneer in antiviral nanomedicines at this Conference. Dr. Diwan has been invited to participate on the panel on "Nanomedicine: Innovation, Partnership and Investment", chaired by Dr. Mostafa Analoui, PhD, Head of Healthcare and Life Sciences, The Livingston Group. The Company plans to include the late-breaking information discussed in this press release in the presentation.
About NanoViricides:
NanoViricides, Inc. (Welcome to NanoViricides, Inc.) is a development stage company that is creating special purpose nanomaterials for viral therapy. The Company's novel nanoviricide(R) class of drug candidates are designed to specifically attack enveloped virus particles and to dismantle them. The Company is developing drugs against a number of viral diseases including H1N1 swine flu, H5N1 bird flu, seasonal Influenza, HIV, oral and genital Herpes, viral diseases of the eye including EKC and herpes keratitis, Hepatitis C, Rabies, Dengue fever, and Ebola virus, among others.
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
playars
lotto players
- Registrato
- 11/4/08
- Messaggi
- 8.306
- Punti reazioni
- 174
NanoViricides Raises an Additional $5 Million in Shelf Offering - The Company Reports Having More than 24 Months of Cash in Hand
announced today that it has raised $5,000,000, drawing down on its previously announced universal registered shelf “Form S-3” offering. The registered shelf offering became effective on April 29, 2010 and continues to remain effective.
The Company received this financing from a single investor, Seaside 88, LP (“Seaside”), a Florida limited partnership. Seaside has previously financed NanoViricides with a total of approximately $10 million under a similar arrangement. Seaside has also financed several leading-edge bio-pharma companies, including Cytori Therapeutics, Inc., Generex Biotechnology Corporation, and NovaDel Pharma, Inc. among others.
NanoViricides has entered into a securities purchase agreement with Seaside for the purchase and sale of up to 500,000 shares of its Series B Preferred Stock at the purchase price of $10.00 per share. On April 19, 2011 Seaside purchased an initial 250,000 shares of the Company’s Series B Preferred Stock at the purchase price of $10.00 per share for an aggregate purchase price of $2,500,000. The Company received $2.5M upon closing, with a net of approximately $2.30M after deducting brokerage commission and expenses. The first conversion of $400,000 of Series B Preferred stock to common stock took place on April 19, 2011. Additional conversions will follow every fourteen days. The conversion price will be based on the same terms and conditions as the previous financing the Company concluded with Seaside (described below).
“We are pleased with the continuing support and confidence in the Company that Seaside has demonstrated with this investment,” said Anil R. Diwan, PhD, President of the Company, adding, “We are now in a better financial state in terms of being able to take our drug development programs towards the clinical studies stage.”
“We continue to have more than 24 months of cash in hand, based on current rate of expenditure. This additional investment will enable us to engage into expanding our pre-clinical studies towards filing an IND application to the FDA,” said Eugene Seymour, MD, MPH, CEO of the Company.
The Series B Preferred Stock is convertible into a number of shares of the Company’s common stock every two weeks. The converted shares are estimated to represent less than five percent of the 10-day trading volume of the Company’s stock (NNVC : OTC-BB), based upon current data.
Of the shares purchased, 40,000 shares of the Series B Preferred Stock will be automatically converted into common stock every two weeks beginning April 19, 2011. The conversion factor shall equal the purchase price of $10 per share of the preferred stock, divided by the lesser of (i) the ten day daily volume weighted average of actual trading prices (“VWAP”) of the common stock multiplied by 0.85; or (ii) the VWAP for the trading day immediately prior to a conversion date multiplied by 0.88. In addition, the unconverted shares of the Series B Preferred Stock will accrue a dividend at a 10% annualized rate. The accrued dividend shall be payable in common stock at the time of each conversion. The Company does not pay a dividend on the shares of its common stock or the shares of its Preferred Series A stock, and will not be able to pay any dividend on these securities while any shares of the Series B Preferred stock remain unconverted. The shares of Series B Preferred Stock and the shares of common stock underlying the Series B Preferred Stock and the dividend earned on it were offered pursuant to an effective shelf registration statement. The Series B Preferred Stock does not have any voting rights except as set forth in the Certificate of Designation, as amended, creating the stock.
Midtown Partners & Co., LLC, acted as the placement agent for this transaction. Midtown received a cash placement fee of 6%.
A shelf registration statement relating to the shares of common stock underlying the shares of preferred stock issued in the offering has been filed with the Securities and Exchange Commission (the “SEC”) and has been declared effective. A prospectus supplement relating to the current transaction has been filed by NanoViricides with the SEC. Copies of the prospectus supplement and accompanying prospectus may be obtained directly from NanoViricides by contacting NanoViricides, Inc., 135 Wood Street, Suite 205, West Haven, Connecticut 06516. This announcement is neither an offer to sell nor a solicitation of an offer to buy any shares of preferred or common stock of NanoViricides. No offer, solicitation or sale will be made in any jurisdiction in which such offer, solicitation or sale is unlawful.
About NanoViricides:
NanoViricides, Inc. (Welcome to NanoViricides, Inc.) is a development stage company that is creating special purpose nanomaterials for antiviral therapy. The Company's novel nanoviricide® class of drug candidates are designed to specifically attack enveloped virus particles and to dismantle them. The Company is developing drugs against a number of viral diseases including H1N1 swine flu, H5N1 bird flu, seasonal Influenza, HIV, oral and genital Herpes (HSV), viral diseases of the eye including EKC and herpes keratitis, Hepatitis C, Rabies, Dengue fever, and Ebola virus, among others.
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
per questa grande news
NanoViricides, Inc. Files Quarterly Report - Reports a Strong Cash Position Sufficient for Phase I Clinical Trials of its First Drug Candidate
hanno cassa per due anni
zero debiti
$13.880M in cash
il resto è scritto tutto qua:
NanoViricides, Inc. Files Quarterly Report - Reports a Strong Cash Position Sufficient for Phase I Clinical Trials of its First Drug Candidate - Yahoo! Finance
Allegati
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419