Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Ocugen OCGN ( ex HSGX )
- Creatore Discussione oroly
- Data di inizio
Più opzioni
Chi ha risposto?Histogenics Corporation CEO to present at conference
http://www.metrowestdailynews.com/news/20160729/histogenics-corporation-ceo-to-present-at-conference
http://www.metrowestdailynews.com/news/20160729/histogenics-corporation-ceo-to-present-at-conference
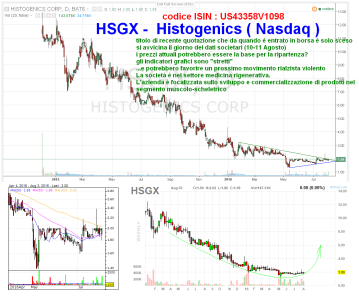
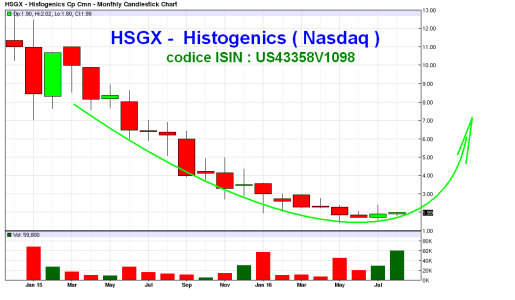
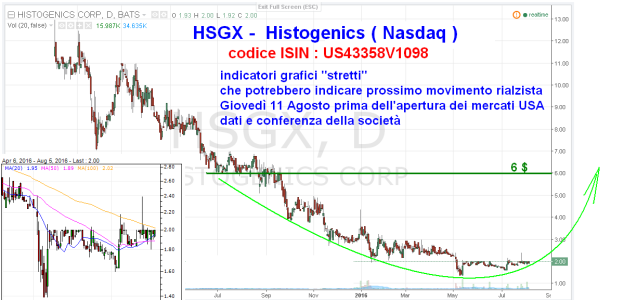
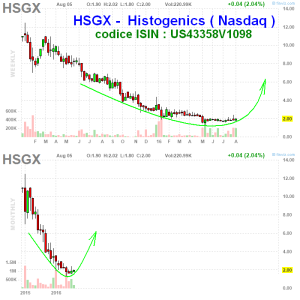
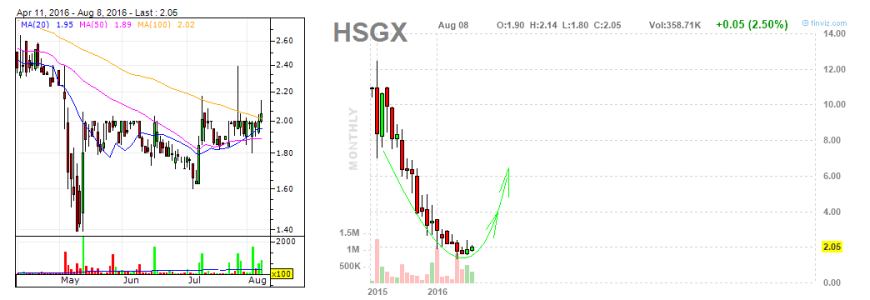
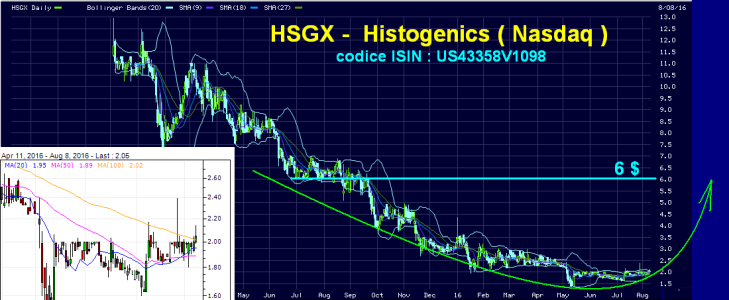
questa è pronta per sbombolare al rialzo

http://www.finanzaonline.com/forum/wall-street/1764225-hsgx-histogenics-nasdaq.html?highlight=hsgx
ma sei scemo oppure rinco?
ti ho appena scritto di usare la funz cerca
http://www.finanzaonline.com/forum/wall-street/1764225-hsgx-histogenics-nasdaq.html?highlight=hsgx
ma sei scemo oppure rinco?
ti ho appena scritto di usare la funz cerca
il nick "cicciobello5 " mi sta un pò antipatico... per questo ho preferito farne 1 nuovo...
anche xchè lì c'era solo 1 post mi pare...
( chiedo ai moderatori magari di unire quell'unico post... in questa discussione )
pure questo?il nick "cicciobello5 " mi sta un pò antipatico... per questo ho preferito farne 1 nuovo...
anche xchè lì c'era solo 1 post mi pare...
( chiedo ai moderatori magari di unire quell'unico post... in questa discussione )
http://www.finanzaonline.com/forum/...-biotec-della-cartilagine.html?highlight=hsgx
che poi magari non producesse fuffa mi servirebbero i suoi farmaci
davicerr
Nuovo Utente
- Registrato
- 23/4/09
- Messaggi
- 4.942
- Punti reazioni
- 70
Interessante, penso che arriverà anche il suo turno. Ps mi dice che il tuo utente non è abilitato a ricevere messaggi privati..forse li hai disabilitati per errori
Zeropercento
fedele alla linea
- Registrato
- 12/1/06
- Messaggi
- 10.990
- Punti reazioni
- 543
- Registrato
- 8/8/16
- Messaggi
- 91
- Punti reazioni
- 3
Histogenics Corporation Announces Second Quarter 2016 Financial and Operating Results
- NeoCart® Phase 3 Clinical Trial Remains on Track for Enrollment
Completion by End of Second Quarter of 2017 -
- Increasing Year-End 2016 Enrollment Guidance to 190 to 200 patients -
- Company to Host Conference Call and Webcast Today at 8:30 a.m. EDT -
WALTHAM, Mass., Aug. 11, 2016 (GLOBE NEWSWIRE) -- Histogenics Corporation (Histogenics) (Nasdaq:HSGX), a regenerative medicine company focused on developing and commercializing products in the musculoskeletal space, announced its financial and operational results for the quarter ended June 30, 2016.
“We are very pleased with the progress we have made during the first half of 2016. Our underlying business fundamentals continue to strengthen, with enrollment ahead of plan and our continued transition to fully integrated manufacturing in support of our anticipated BLA filing with the FDA,” stated Adam Gridley, President and Chief Executive Officer of Histogenics. “With our recent operational success, we continue to narrow our focus on upcoming near-term milestones, including enrollment progress, an interim analysis in early 2017 and continued progress on our pipeline activities in Japan and with our partner Intrexon. We will also continue to explore potential commercial partnering alternatives for NeoCart that may enable us to further expand this important therapy outside of the U.S.,” continued Mr. Gridley.
Second Quarter 2016 and Recent Highlights
NeoCart Phase 3 Clinical Trial Status: As of August 10, 2016 Histogenics has enrolled 167 of the 245 patients required under the Special Protocol Assessment (SPA) with the United States Food and Drug Administration (FDA) in its NeoCart Phase 3 clinical trial. Following a strong start to the year, recent increasing enrollment trends continue to run ahead of Histogenics’ expectations. As a result, Histogenics is narrowing its year-end enrollment guidance by increasing the low end of the range from 180 to 190 patients. In addition, Histogenics confirms its expectations that patient enrollment will be complete by the end of the second quarter of 2017. There are 35 sites participating in the clinical trial as of August 10, 2016.
NeoCart Product Rights for Japanese Market: Histogenics acquired the NeoCart development and commercialization rights for the Japanese market from its long-time, development partner Purpose Co., Ltd (Purpose). Histogenics intends to capitalize on the recent advancements in regenerative medicine regulatory pathways in Japan and its robust Phase 1 and 2 data packages. Additionally, Histogenics is re-engaging with Japanese regulatory authorities with the next informal and formal meetings targeted for the second half of 2016. In parallel, Histogenics is engaging with development and commercialization partners in Japan and elsewhere in Asia with a goal of securing commercial partners after receiving clarity on the path to commercialization from the Japanese regulatory authorities.
Histogenics Raw Materials Approved by FDA and Incorporated into NeoCart Phase 3 Trial: Histogenics reached agreement with the FDA in April 2016 regarding the development and transition of the production of certain critical raw materials for NeoCart from third party suppliers to its in-house manufacturing facility in Lexington, Massachusetts. This is another milestone in a project that was initiated in 2013 with several goals, including improving the quality of critical raw materials, lowering the future cost of goods sold and ensuring ample supply to support both the ongoing clinical trial and the potential future commercial launch of NeoCart. In 2015, Histogenics completed the qualification runs for its collagen, a key raw material needed for the manufacture of NeoCart with submission of the supporting data to the FDA in March of 2016. In April 2016, the FDA approved the use of the internally produced collagen and Histogenics began using this material in the ongoing Phase 3 trial in June 2016.
Intrexon Collaboration: Histogenics and Intrexon Corporation (Intrexon), Histogenics’ collaboration partner for potential next generation products, continued to advance their collaboration for the development of an iPSC chondrocyte program. The partners held a meeting of medical, regulatory and scientific experts to both evaluate the analytical and comparability data generated since the beginning of 2016 and determine the most efficient development plan and regulatory pathway. The partners are currently using feedback from the meeting to determine the need for any additional work prior to engaging with the FDA and other regulatory authorities and anticipate the identification of a development plan prior to the end of 2016.
“We have now enrolled approximately two-thirds of the 245 patients required to complete the NeoCart Phase 3 clinical trial under the SPA. Based on the recent positive enrollment trend, we are increasing the bottom range of our enrollment guidance to 190 patients for revised guidance of 190 to 200 patients enrolled in the trial by the end of 2016,” stated Mr. Gridley.
Financial Results for the Second Quarter of 2016
For the second quarter of 2016, Histogenics reported a net loss attributable to common stockholders of $(8.0) million, or $(0.61) per share, compared to $(7.6) million, or $(0.58) per share, in the second quarter of 2015.
Research and development expenses were $5.8 million in the second quarter of 2016, compared to $5.9 million in the second quarter of 2015. The decrease in expense was primarily due to raw materials purchases in the second quarter of 2015 to support the NeoCart Phase 3 clinical trial combined with a reduction in consulting expense in the second quarter of 2016. This decrease was partially offset by an increase in headcount and clinical trial related expenses in the second quarter of 2016. General and administrative expenses were $2.2 million in the second quarter of 2016, compared to $1.7 million in the second quarter of 2015. The increase was primarily due to higher salaries, facility-related and legal costs which were partially offset by a reduction in consulting expense.
At June 30, 2016, Histogenics had cash, cash equivalents and marketable securities of $15.9 million, compared to $30.9 million at December 31, 2015. Histogenics believes its current cash position will fund its operations into the first quarter of 2017.
fonte : http://www.einnews.com/pr_news/3394...-quarter-2016-financial-and-operating-results
- NeoCart® Phase 3 Clinical Trial Remains on Track for Enrollment
Completion by End of Second Quarter of 2017 -
- Increasing Year-End 2016 Enrollment Guidance to 190 to 200 patients -
- Company to Host Conference Call and Webcast Today at 8:30 a.m. EDT -
WALTHAM, Mass., Aug. 11, 2016 (GLOBE NEWSWIRE) -- Histogenics Corporation (Histogenics) (Nasdaq:HSGX), a regenerative medicine company focused on developing and commercializing products in the musculoskeletal space, announced its financial and operational results for the quarter ended June 30, 2016.
“We are very pleased with the progress we have made during the first half of 2016. Our underlying business fundamentals continue to strengthen, with enrollment ahead of plan and our continued transition to fully integrated manufacturing in support of our anticipated BLA filing with the FDA,” stated Adam Gridley, President and Chief Executive Officer of Histogenics. “With our recent operational success, we continue to narrow our focus on upcoming near-term milestones, including enrollment progress, an interim analysis in early 2017 and continued progress on our pipeline activities in Japan and with our partner Intrexon. We will also continue to explore potential commercial partnering alternatives for NeoCart that may enable us to further expand this important therapy outside of the U.S.,” continued Mr. Gridley.
Second Quarter 2016 and Recent Highlights
NeoCart Phase 3 Clinical Trial Status: As of August 10, 2016 Histogenics has enrolled 167 of the 245 patients required under the Special Protocol Assessment (SPA) with the United States Food and Drug Administration (FDA) in its NeoCart Phase 3 clinical trial. Following a strong start to the year, recent increasing enrollment trends continue to run ahead of Histogenics’ expectations. As a result, Histogenics is narrowing its year-end enrollment guidance by increasing the low end of the range from 180 to 190 patients. In addition, Histogenics confirms its expectations that patient enrollment will be complete by the end of the second quarter of 2017. There are 35 sites participating in the clinical trial as of August 10, 2016.
NeoCart Product Rights for Japanese Market: Histogenics acquired the NeoCart development and commercialization rights for the Japanese market from its long-time, development partner Purpose Co., Ltd (Purpose). Histogenics intends to capitalize on the recent advancements in regenerative medicine regulatory pathways in Japan and its robust Phase 1 and 2 data packages. Additionally, Histogenics is re-engaging with Japanese regulatory authorities with the next informal and formal meetings targeted for the second half of 2016. In parallel, Histogenics is engaging with development and commercialization partners in Japan and elsewhere in Asia with a goal of securing commercial partners after receiving clarity on the path to commercialization from the Japanese regulatory authorities.
Histogenics Raw Materials Approved by FDA and Incorporated into NeoCart Phase 3 Trial: Histogenics reached agreement with the FDA in April 2016 regarding the development and transition of the production of certain critical raw materials for NeoCart from third party suppliers to its in-house manufacturing facility in Lexington, Massachusetts. This is another milestone in a project that was initiated in 2013 with several goals, including improving the quality of critical raw materials, lowering the future cost of goods sold and ensuring ample supply to support both the ongoing clinical trial and the potential future commercial launch of NeoCart. In 2015, Histogenics completed the qualification runs for its collagen, a key raw material needed for the manufacture of NeoCart with submission of the supporting data to the FDA in March of 2016. In April 2016, the FDA approved the use of the internally produced collagen and Histogenics began using this material in the ongoing Phase 3 trial in June 2016.
Intrexon Collaboration: Histogenics and Intrexon Corporation (Intrexon), Histogenics’ collaboration partner for potential next generation products, continued to advance their collaboration for the development of an iPSC chondrocyte program. The partners held a meeting of medical, regulatory and scientific experts to both evaluate the analytical and comparability data generated since the beginning of 2016 and determine the most efficient development plan and regulatory pathway. The partners are currently using feedback from the meeting to determine the need for any additional work prior to engaging with the FDA and other regulatory authorities and anticipate the identification of a development plan prior to the end of 2016.
“We have now enrolled approximately two-thirds of the 245 patients required to complete the NeoCart Phase 3 clinical trial under the SPA. Based on the recent positive enrollment trend, we are increasing the bottom range of our enrollment guidance to 190 patients for revised guidance of 190 to 200 patients enrolled in the trial by the end of 2016,” stated Mr. Gridley.
Financial Results for the Second Quarter of 2016
For the second quarter of 2016, Histogenics reported a net loss attributable to common stockholders of $(8.0) million, or $(0.61) per share, compared to $(7.6) million, or $(0.58) per share, in the second quarter of 2015.
Research and development expenses were $5.8 million in the second quarter of 2016, compared to $5.9 million in the second quarter of 2015. The decrease in expense was primarily due to raw materials purchases in the second quarter of 2015 to support the NeoCart Phase 3 clinical trial combined with a reduction in consulting expense in the second quarter of 2016. This decrease was partially offset by an increase in headcount and clinical trial related expenses in the second quarter of 2016. General and administrative expenses were $2.2 million in the second quarter of 2016, compared to $1.7 million in the second quarter of 2015. The increase was primarily due to higher salaries, facility-related and legal costs which were partially offset by a reduction in consulting expense.
At June 30, 2016, Histogenics had cash, cash equivalents and marketable securities of $15.9 million, compared to $30.9 million at December 31, 2015. Histogenics believes its current cash position will fund its operations into the first quarter of 2017.
fonte : http://www.einnews.com/pr_news/3394...-quarter-2016-financial-and-operating-results
Zeropercento
fedele alla linea
- Registrato
- 12/1/06
- Messaggi
- 10.990
- Punti reazioni
- 543
mmm
Zeropercento
fedele alla linea
- Registrato
- 12/1/06
- Messaggi
- 10.990
- Punti reazioni
- 543