Carlito Brigante
Nuovo Utente
- Registrato
- 3/6/08
- Messaggi
- 998
- Punti reazioni
- 29
ottima news natalizia!!! 


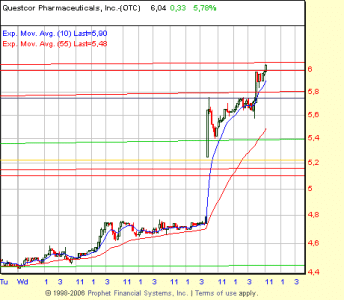
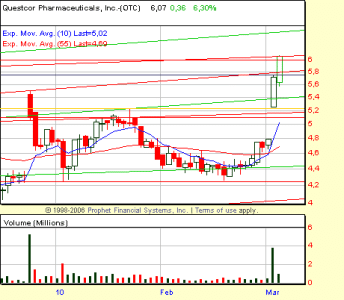
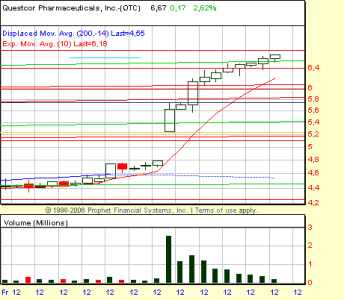
di conseguenza, fiammata con apertura a 5.45, max 5.83 e chiusura a 5.10
stiamo all'occhio alla riapertura. Buon Natale!!!
FDA Accepts for Review Questcor's Filing of Its Supplemental New Drug Application for H.P. Acthar(R) Gel for Treatment of Infantile Spasms
UNION CITY, Calif., Dec. 24, 2009 (GLOBE NEWSWIRE) -- Questcor Pharmaceuticals, Inc. (Nasdaq:QCOR) today announced the U.S. Food & Drug Administration (FDA) now considers that Questcor has provided a complete response to all prior action letters for its supplemental New Drug Application (sNDA) to market H.P. Acthar(R) Gel (repository corticotrophin injection) for the treatment of infantile spasms. The FDA has set the user fee goal date (PDUFA) of June 11, 2010 for this sNDA.
"The medical community and Questcor continue to believe that Acthar has an important role in the treatment of infantile spasms and we look forward to working with the FDA to answer any questions as they consider our application," said Don M. Bailey, President and CEO of Questcor. "We continue to expect the FDA will convene an Advisory Panel meeting to obtain independent expert advice on specific aspects of the sNDA," added Mr. Bailey.
Acthar is currently approved in the U.S. for the treatment of MS exacerbations, nephrotic syndrome and many other conditions. Acthar is not approved in the U.S. for the treatment of IS, a potentially life-threatening disorder that typically begins in the first year of life. However, pursuant to guidelines published by the American Academy of Neurology and the Child Neurology Society, many child neurologists use Acthar to treat infants afflicted with this condition. Previously, the FDA granted Orphan Designation to Acthar for the treatment of IS.
About Questcor
Questcor Pharmaceuticals, Inc. is a pharmaceutical company that markets H.P. Acthar(R) Gel (repository corticotropin injection). H.P. Acthar Gel ("Acthar") is an injectable drug that is approved for the treatment of certain disorders with an inflammatory component, including the treatment of exacerbations associated with multiple sclerosis ("MS") and to induce a diuresis or a remission of proteinuria in the nephrotic syndrome without uremia of the idiopathic type or that is due to lupus erythamatosus. In addition, Acthar is not indicated for, but is used in treating patients with infantile spasms ("IS"), a rare form of refractory childhood epilepsy, and opsoclonus myoclonus syndrome, a rare autoimmune-related childhood neurological disorder. The Company also markets Doral(R) (quazepam), which is indicated for the treatment of insomnia characterized by difficulty in falling asleep, frequent nocturnal awakenings, and/or early morning awakenings. For more information, please visit www.questcor.com.

di conseguenza, fiammata con apertura a 5.45, max 5.83 e chiusura a 5.10
stiamo all'occhio alla riapertura. Buon Natale!!!
FDA Accepts for Review Questcor's Filing of Its Supplemental New Drug Application for H.P. Acthar(R) Gel for Treatment of Infantile Spasms
UNION CITY, Calif., Dec. 24, 2009 (GLOBE NEWSWIRE) -- Questcor Pharmaceuticals, Inc. (Nasdaq:QCOR) today announced the U.S. Food & Drug Administration (FDA) now considers that Questcor has provided a complete response to all prior action letters for its supplemental New Drug Application (sNDA) to market H.P. Acthar(R) Gel (repository corticotrophin injection) for the treatment of infantile spasms. The FDA has set the user fee goal date (PDUFA) of June 11, 2010 for this sNDA.
"The medical community and Questcor continue to believe that Acthar has an important role in the treatment of infantile spasms and we look forward to working with the FDA to answer any questions as they consider our application," said Don M. Bailey, President and CEO of Questcor. "We continue to expect the FDA will convene an Advisory Panel meeting to obtain independent expert advice on specific aspects of the sNDA," added Mr. Bailey.
Acthar is currently approved in the U.S. for the treatment of MS exacerbations, nephrotic syndrome and many other conditions. Acthar is not approved in the U.S. for the treatment of IS, a potentially life-threatening disorder that typically begins in the first year of life. However, pursuant to guidelines published by the American Academy of Neurology and the Child Neurology Society, many child neurologists use Acthar to treat infants afflicted with this condition. Previously, the FDA granted Orphan Designation to Acthar for the treatment of IS.
About Questcor
Questcor Pharmaceuticals, Inc. is a pharmaceutical company that markets H.P. Acthar(R) Gel (repository corticotropin injection). H.P. Acthar Gel ("Acthar") is an injectable drug that is approved for the treatment of certain disorders with an inflammatory component, including the treatment of exacerbations associated with multiple sclerosis ("MS") and to induce a diuresis or a remission of proteinuria in the nephrotic syndrome without uremia of the idiopathic type or that is due to lupus erythamatosus. In addition, Acthar is not indicated for, but is used in treating patients with infantile spasms ("IS"), a rare form of refractory childhood epilepsy, and opsoclonus myoclonus syndrome, a rare autoimmune-related childhood neurological disorder. The Company also markets Doral(R) (quazepam), which is indicated for the treatment of insomnia characterized by difficulty in falling asleep, frequent nocturnal awakenings, and/or early morning awakenings. For more information, please visit www.questcor.com.