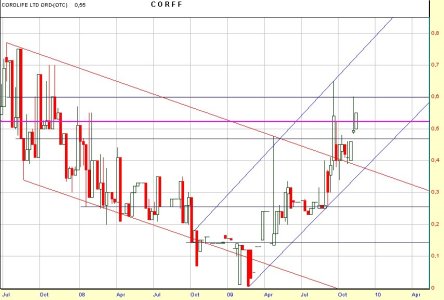
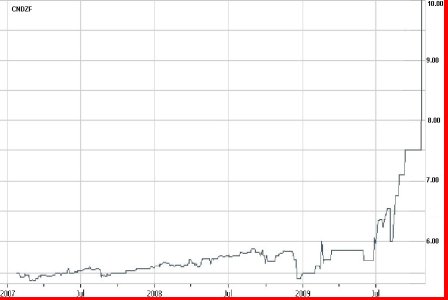
Corff
CORFF
http://www.cordlife.com/
CordLife operates Australasia's largest network of private cord blood banks with full processing and cryopreservation storage facilities in Singapore, Hong Kong, Indonesia and India as well as marketing presence throughout the region. CordLife is listed on the Australian Stock Exchange as 'CBB'. In a short span of 8 years, CordLife has grown to become the leading cord blood bank which over 24,0001 families chose to trust.
CordLife collects, processes and stores cord blood stem cells which may later become potential source material for lifesaving treatment. We provide families with high quality cord blood processing and storage services to protect their children's cord blood stem cells. Cord blood has become a major source of stem cells for transplantation worldwide since the first transplant in 1988. Doctors have used cord blood stem cells to treat more than 14,000* patients suffering from over 80 diseases including certain cancers and bone marrow failure syndromes, inborn errors of metabolism, blood disorders and immunodeficiencies.
Since our inception in 2001, we have established a rigorous quality system and track record of clinically reliable cord blood banking service that was first certified by Singapore's Ministry of Health, and subsequently accredited in 2005 by the world's most recognised quality standards in cord blood banking - American Association of Blood Banks (AABB).
Cordlife Group of companies all over Asia have also attained numerous accreditations and certifications from other quality organisations such as the Therapeutic Goods Administration (TGA), International Organization for Standardization (ISO), and local regulators such as the Ministry of Health (Indonesia) and Ministry of Health (India) to give our clients the added assurance that their baby's cord blood will be collected, processed and stored according to stringent quality standards to deliver safe stem cells for medical treatment. Our ability to obtain various local and international quality marks and regulatory certifications is testimony of the company's intimate knowledge of the cord blood banking industry and its needs.
In 2006, CordLife was awarded the prestigious 'Technology Pioneer' status by the World Economic Forum for advancing the field of adult stem cell therapy, cord blood banking and technologies. CordLife was one of the only 3 companies in Asia to be awarded this rare recognition.
1 Figure accurate as of 28 February 2009.
* Cord Blood Forum. Accessed 10 November 2009
Singapore
About CordLife Pte. Ltd.
Founded in 2001 and certified by the Ministry of Health, CordLife is the first private cord blood bank in Singapore, and among the first in Asia. In 2005, CordLife Singapore achieved the prestigious American Association of Blood Banks (AABB) accreditation for its cord blood banking facility in Camden Medical Centre.
Due to rapid growth, CordLife relocated to a larger facility in Singapore Science Park II. The new facility was successfully reaccredited by AABB in 2008, and today CordLife remains the only AABB accredited cord blood bank and the largest laboratory in Southeast Asia.
As headquarters of the CordLife Group, CordLife Singapore always strives to be a 'Technology Pioneer' by introducing numerous industry firsts. Most recently, we are the first in Asia and Singapore to offer the latest standard in automated cord blood processing technology, known as Sepax®.
Australia
About BioCell Pty. Ltd. (renamed as 'Australian Stem Cell Healthcare')
Incorporated in 2004 by scientists, doctors and nurses, the BioCell team comprise over 25 years of experience in specialised medicine and scientific research such as Haematology, Blood Banking, Immunology and Stem Cell Transplantation. Medical Director of BioCell, Associate Professor Mark Kirkland conducts research on cord blood and collaborates with a range of stem cell biotechnology companies and universities worldwide regards to possible future cellular therapies. He was recently awarded the title 'Researcher of the Year' in Barwon (Australia), for his pioneering work in redirecting the differentiation of stem cells into heart cells. BioCell is licensed by Australia's Therapeutic Goods Administration (TGA). Additionally, the laboratory also adheres to world class standards in line with FACT-Netcord guidelines, one of the highest international standards for blood banking centres.
BioCell has been renamed as 'Australian Stem Cell Healthcare' on 19 March 2009 after merging with CellSense to become the country's largest private cord blood bank.
About Cytomatrix® Pty. Ltd.
Associate Professor Mark Kirkland is also the Chief Scientific Officer of Cytomatrix® Pty. Ltd., a stem cell research company based in Australia. Since May 1996, Cytomatrix® has been successful in developing cell based applications through its patented three-dimensional growth scaffold. It specialises in Ex Vivo Stem Cell Expansion and T Cell Immuno-therapeutics, which are key for development of regenerative medicine. Cytomatrix® is actively involved in scientific collaborations and has established relationships with leading research institutions, hospitals and federal government bodies in the United States.
Indonesia
About P.T. CordLife Indonesia
P.T. CordLife Indonesia remains the only licensed private cord blood banking facility in Indonesia with comprehensive processing and storage capabilities for up to 30,000 cord blood units. Strategically positioned to serve the whole of Indonesia, CordLife?s main office and storage facility are located in Jakarta, with sales offices in Medan and Surabaya.
Officially launched by Health Minister RI Siti Fadillah Supari on 1 October 2007, the facility was built according to the standards set by the American Association of Blood Banks (AABB). The laboratory and storage areas are protected with 24 hour security, uninterrupted power as well as flood and fireproof features.
P.T. CordLife Indonesia is a joint venture between CordLife Ltd. and pharmaceutical giant P.T. Kalbe Farma Tbk.
India
About CordLife Sciences India Pvt. Ltd.
CordLife India is the largest and most advanced cord blood facility in the country, with a storage capacity of up to 150,000 cord blood units. The state-of-the-art laboratory with full processing, testing and cryopreservation capabilities is the latest addition to our group's network of facilities. It is also equipped with continuous power back-up as well as the most-up-to-date security and surveillance systems. In addition, the building is specially designed to withstand any natural calamity.
Singapore Senior Minister and former Prime Minister Mr Goh Chok Tong officially launched CordLife Sciences India on 20 January 2006. Located in Kolkata, CordLife is the only such facility serving the Eastern part of India. We are licensed by the Drug Controller General of India to collect cord blood units from all Indian states and cities.
Hong Kong
About CordLife (Hong Kong) Limited
Launched in March 2005, CordLife Hong Kong is our first overseas facility which was built in line with the standards set by the American Association of Blood Banks (AABB). Within two years, the company was internationally recognised by the International Organization for Standardization with ISO 9001:2000 certification. In 2008, a second office was opened in Kowloon, strategically opposite Hong Kong Baptist Hospital which holds the record for the city's highest number of private deliveries.
Philippines
About CordLife Medical Phils, Inc
CordLife Philippines is our sales and marketing office which conducts cord blood educational and marketing activities for both expectant parents delivering in Philippines. Working in tandem with doctors from various clinics and hospitals, CordLife organises many outreach programmes about the benefits of cord blood stem cells.
Our friendly and professional staffs are on call 24 hours a day, 7 days a week, to assist expectant parents who wish to enroll and store their baby's cord blood stem cells in CordLife Singapore's AABB accredited facility. CordLife is the only AABB accredited private cord blood bank in Philippines to offer Sepax®, the world's preferred automated cord blood processing.
Thailand
About Cygenics (Thailand) Co. Ltd
Operating since March 2005, our sales and marketing office is conveniently located in central Bangkok to serve expectant families in Thailand. In addition to various marketing and support activities, CordLife Thailand works in close partnership with 3 prominent private hospitals which account for a major percentage of deliveries in the country.
CordLife remains the first and only AABB accredited private cord blood bank to offer automated cord blood processing with Sepax®. CordLife works closely with clients and hospitals to facilitate the shipping of cord blood for processing and storage in CordLife Singapore's AABB accredited facility. CordLife is the preferred cord blood bank for many of the expatriates in Thailand.
http://www.cordlife.com/investors/aboutus/intellectualproperties.php
Patent
US Patent 5,282,861
European Patent specification EP 0,560,279B1
The patents cover porous, three-dimensional devices made using a carbon skeleton with a metal coating and further protect the use of such devices for cell growth and expansion.
US patent number 6,440,734
"Methods and Devices for the Long-Term Culture of Progenitor Cells"
The broad patents/applications cover the growth and expansion of blood stem cells (from marrow, peripheral blood and cord blood) in three dimensional devices.
US patent number 6,548,299
China Patent ZL 98 8 13230.6
"Lymphoid Tissue-Specific Cell Production From Hematopoietic Progenitor Cells In Three-Dimensional Devices"
The broad patents/applications cover the production of a broad range of immune cells ex vivo in three-dimensional devices.
Patents Pending
"Cell Culture Spinner Flasks"
Series of pending patents covering the rotational spinner flask culture system for high cell growth performance.
"Methods and Devices for Obtaining Non-Hematopoietic Lineage Cells from Hematopoietic Progenitor Cells"
Pending patents covering a novel, multipotent stem cell isolated from blood/marrow that exhibits trans-differentiation capabilities into non-hematopoietic tissue, such as skin (epithelium), vessels (endothelium) and neurons.
"Cytokine-Free Growth and Maintenance of Progenitor Cells"
First-stage filing covering the growth and expansion of blood stem cells in media without cytokine augmentation.
"Cell Culture System"
Patent applications covering a combination of biocompatible 3-D cell culture scaffold and membrane technology yielding cell expansion.
http://www.reuters.com/finance/stocks/companyProfile?symbol=CORFF.PK
http://www.reuters.com/finance/stocks/ratios?symbol=CORFF.PK
Cordlife Limited is an Australia-based company. The Company and its subsidiaries are engaged in the provision of cord blood banking services, which involves the processing and storage of stem cells. It operates in Australia and Asia. It collects, processes and stores baby's cord blood stem cells. It has established network of private cord blood banks with processing and storage facilities in Singapore, Hong Kong, Indonesia and India as well as marketing operations in Philippines, Thailand and Macau. Its subsidiaries include Cordlife (Hong Kong) Ltd, Cordlife Sciences Ltd, Cryogenics (Thailand) Ltd, Cordlife Medical Phils Inc. and CLS Services B.V. As at June 30, 2009, the Company’s total client numbers was approximately 21,000. On December 29, 2008, Biocell Pty Ltd, previously 57%-owned subsidiary of Cordlife Ltd, merged with CellSense Pty Ltd.