Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
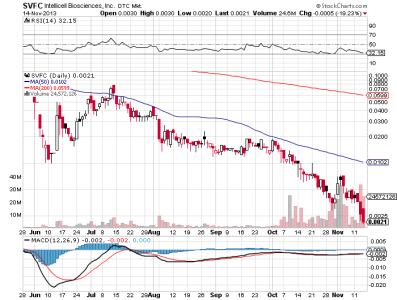
svfc
- Creatore Discussione axelvento
- Data di inizio
Più opzioni
Chi ha risposto?visal
In cul@ al nasdaq
- Registrato
- 20/6/01
- Messaggi
- 23.858
- Punti reazioni
- 304
Chi non muore si rivede.0136
ipervenduto
biotec
Ciao socio tra breve trattero' le ciofete
Ahahhahahah

axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
Chi non muore si rivede
Ciao socio tra breve trattero' le ciofete
Ahahhahahah
ciao
primario
come stai..la salute????
come mai tratti le ciofeche?
hai picchiato la testa
visal
In cul@ al nasdaq
- Registrato
- 20/6/01
- Messaggi
- 23.858
- Punti reazioni
- 304
Sembra che siano gli unici titoli x perdere tutto e subito e toglierti il pensierociao
primario
come stai..la salute????
come mai tratti le ciofeche?
hai picchiato la testa


axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
Sembra che siano gli unici titoli x perdere tutto e subito e toglierti il pensiero
o tutto o niente...

visal
In cul@ al nasdaq
- Registrato
- 20/6/01
- Messaggi
- 23.858
- Punti reazioni
- 304
Bravo o ricco pescatore o povero marinaio-
o tutto o niente...
Io voglio diventare ricco pescatore



axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
Bravo o ricco pescatore o povero marinaio
Io voglio diventare ricco pescatore
allora vai a comprare la canna da pesca..
e spera che abbocchi qualcosa
visal
In cul@ al nasdaq
- Registrato
- 20/6/01
- Messaggi
- 23.858
- Punti reazioni
- 304
Voglio pescare le ciofeteallora vai a comprare la canna da pesca..
e spera che abbocchi qualcosa
Ahahha
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
.0136
ipervenduto
biotec
.017
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
colpo secco!
M 2.7
.012
picchiamo pesante
$ 0,014 64,000 OTO 15:46:12
$ 0,014 60,000 OTO 15:32:07
$ 0,014 40,000 OTO 15:32:06
$ 0,014 10,000 OTO 13:08:11
$ 0,014 9,000 OTO 13:08:09
$ 0,014 25,000 OTO 12:55:59
$ 0,0125 78,400 OTO 12:55:53
$ 0,0125 60,000 OTO 12:55:07
$ 0,012 53,000 OTO 12:34:34
$ 0,014 10,000 OTO 11:22:56
$ 0,014 14,000 OTO 11:21:45
$ 0,014 10,000 OTO 11:06:48
$ 0,014 75,000 OTO 11:04:53
$ 0,014 6,000 OTO 11:02:43
$ 0,012 100,000 OTO 10:59:59
$ 0,012 2,700,000 OTO 10:50:00 $32.400
$ 0,0101 136,690 OTO 10:31:29
$ 0,0101 60,000 OTO 10:31:21
$ 0,011 10,000 OTO 10:30:55
$ 0,012 40,000 OTO 10:28:26
$ 0,012 15,000 OTO 10:28:19
$ 0,0121 250,000 OTO 10:28:00
$ 0,0124 170,000 OTO 10:26:24
$ 0,0125 75,000 OTO 10:25:57
$ 0,0127 65,000 OTO 10:21:39
$ 0,0128 40,000 OTO 10:05:30
$ 0,0125 54,000 OTO 10:05:18
$ 0,0127 58,200 OTO 10:05:09
$ 0,0126 500,000 OTO 10:05:07
$ 0,0126 508,000 OTO 09:59:19
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
bella news
ntelliCell Biosciences Stem Cells Used to Successfully Treat Bell's Palsy and Type I Diabetes Patient
NEW YORK, NY--(Marketwired - Oct 9, 2013) - IntelliCell Biosciences, Inc. (OTCQB: SVFC), a regenerative medicine company utilizing adult autologous vascular cells (SVCs) derived from the blood vessels, announced today that it has supplied its cellular product, stromal vascular fraction cells ("SVFC"), to Regen Medical PC for the successful treatment of a Type I diabetic patient, who had developed bilateral permanent Bell's palsy.
Previous medical history stated neither the patient's diabetes nor Bell's palsy had improved following 6 months of multiple congenital treatments. Following the unsuccessful treatments, the patient was remitted to the care of Dr. Steven Victor of ReGen Medical PC. Under the care of Dr. Victor, the patient received two treatments of ReGen Medical's proprietary stem cell applications spaced 6 months apart. The result was a total resolution of the Bell's palsy. The patient also noted that his insulin requirements for his Type I diabetes was reduced from 60 Units of Insulin daily to 5-10 Units of Insulin daily.
Dr. Steven Victor, Chairman and CEO of IntelliCell Biosciences, commented, "This was a clinical treatment utilizing IntelliCell's SVFC method, which addressed an unmet clinical need. If not for our successful treatments, this patient was going to have to undergo corrective surgery of his eye and face and retire from his job due to slurred speech. Prior to our treatment the patient had been treated at some of most respected Medical Centers without getting any positive results. It is rewarding to have been part of a treatment for a patient where our cellular product helped change his quality of life. IntelliCell firmly believes that the future of medicine will entail utilizing cellular therapy for previously unmet clinical needs. Additionally, we were excited to see the patient's insulin requirements reduced and we are looking into the possibility of our cellular product for future treatments of Diabetes Type I and Type II."
Bell's palsy is a form of facial paralysis resulting from a dysfunction of the cranial nerve VII (the facial nerve) causing an inability to control facial muscles on the affected side. Major complications of the condition are chronic loss of taste (ageusia), chronic facial spasms, facial pain and corneal infections.
Diabetes is a group of metabolic diseases in which a person has high blood sugar, either because the pancreas does not produce enough insulin, or because cells do not respond to the insulin that is produced. Type I Diabetes results from the body's failure to produce insulin, and currently requires the person to inject insulin or wear an insulin pump. All forms of diabetes increase the risk of long-term complications. The major long-term complications relate to damage to blood vessels. Diabetes also doubles the risk of cardiovascular disease.
"IntelliCell believes that we possess a paradigm shifting discovery that will change the manner in which patients are treated," Dr. Victor continued. "We are confident that our relationships with the FDA and other governing bodies will allow us to rapidly expand the amounts of patients that we can treat using our SFV therapy."
About IntelliCell Biosciences
IntelliCell is a pioneering regenerative medicine company focused on the expanding regenerative medical markets using adult autologous stromal vascular fraction cells (SVFCs) derived from the blood vessels in the adult adipose tissue. IntelliCell Biosciences has developed its own patented technology and protocol to separate adult autologous vascular cells from adipose tissue without the use of enzymes. IntelliCell will also be seeking to develop technology-licensing agreements with technology developers, universities, and international business entities.
Forward-Looking Statements
Certain statements set forth in this press release constitute "forward-looking statements." Forward-looking statements include, without limitation, any statement that may predict, forecast, indicate, or imply future results, performance or achievements, and may contain the words "estimate," "project," "intend," "forecast," "anticipate," "plan," "planning," "expect," "believe," "will likely," "will reach," "will change," "will soon," "should," "could," "would," "may," "can" or words or expressions of similar meaning. Such statements are not guarantees of future performance and are subject to risks and uncertainties that could cause the company's actual results and financial position to differ materially from those included within the forward-looking statements. Forward-looking statements involve risks and uncertainties, including those relating to the Company's ability to grow its business. Actual results may differ materially from the results predicted and reported results should not be considered as an indication of future performance. The potential risks and uncertainties include, among others, the Company's limited operating history, the limited financial resources, domestic or global economic conditions, activities of competitors and the presence of new or additional competition, and changes in Federal or State laws. More information about the potential factors that could affect the Company's business and financial results is included in the Company's filings, available via the United States Securities and Exchange Commission.
Contacts:
IntelliCell Biosciences, Inc.
Investor Relations
Contact:
Anna Rhodes
Email Contact
(646) 576-8308
ntelliCell Biosciences Stem Cells Used to Successfully Treat Bell's Palsy and Type I Diabetes Patient
NEW YORK, NY--(Marketwired - Oct 9, 2013) - IntelliCell Biosciences, Inc. (OTCQB: SVFC), a regenerative medicine company utilizing adult autologous vascular cells (SVCs) derived from the blood vessels, announced today that it has supplied its cellular product, stromal vascular fraction cells ("SVFC"), to Regen Medical PC for the successful treatment of a Type I diabetic patient, who had developed bilateral permanent Bell's palsy.
Previous medical history stated neither the patient's diabetes nor Bell's palsy had improved following 6 months of multiple congenital treatments. Following the unsuccessful treatments, the patient was remitted to the care of Dr. Steven Victor of ReGen Medical PC. Under the care of Dr. Victor, the patient received two treatments of ReGen Medical's proprietary stem cell applications spaced 6 months apart. The result was a total resolution of the Bell's palsy. The patient also noted that his insulin requirements for his Type I diabetes was reduced from 60 Units of Insulin daily to 5-10 Units of Insulin daily.
Dr. Steven Victor, Chairman and CEO of IntelliCell Biosciences, commented, "This was a clinical treatment utilizing IntelliCell's SVFC method, which addressed an unmet clinical need. If not for our successful treatments, this patient was going to have to undergo corrective surgery of his eye and face and retire from his job due to slurred speech. Prior to our treatment the patient had been treated at some of most respected Medical Centers without getting any positive results. It is rewarding to have been part of a treatment for a patient where our cellular product helped change his quality of life. IntelliCell firmly believes that the future of medicine will entail utilizing cellular therapy for previously unmet clinical needs. Additionally, we were excited to see the patient's insulin requirements reduced and we are looking into the possibility of our cellular product for future treatments of Diabetes Type I and Type II."
Bell's palsy is a form of facial paralysis resulting from a dysfunction of the cranial nerve VII (the facial nerve) causing an inability to control facial muscles on the affected side. Major complications of the condition are chronic loss of taste (ageusia), chronic facial spasms, facial pain and corneal infections.
Diabetes is a group of metabolic diseases in which a person has high blood sugar, either because the pancreas does not produce enough insulin, or because cells do not respond to the insulin that is produced. Type I Diabetes results from the body's failure to produce insulin, and currently requires the person to inject insulin or wear an insulin pump. All forms of diabetes increase the risk of long-term complications. The major long-term complications relate to damage to blood vessels. Diabetes also doubles the risk of cardiovascular disease.
"IntelliCell believes that we possess a paradigm shifting discovery that will change the manner in which patients are treated," Dr. Victor continued. "We are confident that our relationships with the FDA and other governing bodies will allow us to rapidly expand the amounts of patients that we can treat using our SFV therapy."
About IntelliCell Biosciences
IntelliCell is a pioneering regenerative medicine company focused on the expanding regenerative medical markets using adult autologous stromal vascular fraction cells (SVFCs) derived from the blood vessels in the adult adipose tissue. IntelliCell Biosciences has developed its own patented technology and protocol to separate adult autologous vascular cells from adipose tissue without the use of enzymes. IntelliCell will also be seeking to develop technology-licensing agreements with technology developers, universities, and international business entities.
Forward-Looking Statements
Certain statements set forth in this press release constitute "forward-looking statements." Forward-looking statements include, without limitation, any statement that may predict, forecast, indicate, or imply future results, performance or achievements, and may contain the words "estimate," "project," "intend," "forecast," "anticipate," "plan," "planning," "expect," "believe," "will likely," "will reach," "will change," "will soon," "should," "could," "would," "may," "can" or words or expressions of similar meaning. Such statements are not guarantees of future performance and are subject to risks and uncertainties that could cause the company's actual results and financial position to differ materially from those included within the forward-looking statements. Forward-looking statements involve risks and uncertainties, including those relating to the Company's ability to grow its business. Actual results may differ materially from the results predicted and reported results should not be considered as an indication of future performance. The potential risks and uncertainties include, among others, the Company's limited operating history, the limited financial resources, domestic or global economic conditions, activities of competitors and the presence of new or additional competition, and changes in Federal or State laws. More information about the potential factors that could affect the Company's business and financial results is included in the Company's filings, available via the United States Securities and Exchange Commission.
Contacts:
IntelliCell Biosciences, Inc.
Investor Relations
Contact:
Anna Rhodes
Email Contact
(646) 576-8308
axelvento
dCellVax
- Registrato
- 21/12/04
- Messaggi
- 16.246
- Punti reazioni
- 419
news...
.0041
alert$$$$
IntelliCell Biosciences Cellular Therapy Treatment Yields Positive Results in Multiple Sclerosis PatientsFont
8:57 AM ET 11/12/13 | Marketwire
IntelliCell Biosciences, Inc. (OTCQB: SVFC), a regenerative medicine company utilizing adult autologous vascular cells (SVCs) derived from the blood vessels found in adipose tissue, announced today that it has supplied its cellular product, stromal vascular fraction cells ("SVFC"), to ReGen Medical PC for the successful treatment of a patient with Multiple Sclerosis.
It is important to note that by "successful," the company is declaring that these cells do not cure Multiple Sclerosis, but of the 19 patients with MS that ReGen Medical has treated using IntelliCell's technology, each have gained anywhere from good to significant quality of life improvements.
In almost every case that ReGen treated, as well as in this case, previous medical history stated the patients' Multiple Sclerosis had progressively worsened. In the case of this patient she had undergone seven years of progression since her original diagnosis. This patient was remitted to the care of Dr. Steven Victor of ReGen Medical PC, where the patient received six treatments of ReGen Medical's proprietary SVFC, which includes stem cells, spaced approximately 3-5 months apart. The results of the treatment reflected significant improvements to the patient's quality of living. Prior to any SVFC treatments, the patient experienced a gradual decline, beginning in 2001, of her vision, bladder control, and mobility, particularly of the extremities on her right side. This culminated in her diagnosis of stage IV multiple sclerosis in 2006. Following the first treatment, the patient regained use of her previously immobile right leg, and coupled with more treatments and physical therapy, the patient can now walk. After her second treatment, the patient noticed more strength and agility in her previously clenched right hand. Additionally, with further treatment the patient noted drastically improved vision in her previously debilitated eye and she regained bladder control. With each patient ReGen Medical has treated, the number of treatments has varied between one and six, each patient experiencing different improvements at various intervals.
Dr. Steven Victor, CEO of IntelliCell Biosciences, commented, "The patient mentioned in this release, was the first clinical case we treated for MS with IntelliCell's SVFC technology. If not for our beneficial treatments, this patient would be restricted to a wheelchair and lose total sight in one of her eyes. Previously, this patient had been unsuccessfully treated at some of the most respected Medical Centers in the country and received little or no improvement to her symptoms. By utilizing IntelliCell's paradigm shifting medical solutions, we have dramatically improved the quality of this patient's life. IntelliCell is confident that the future of medicine will entail utilizing cellular therapy for previously unmet clinical needs, and we expect to establish ourselves as a market leader in this emerging industry."
Multiple Sclerosis (MS), also known as disseminated sclerosis or encephalomyelitis disseminata, is an inflammatory disease in which the insulating covers, called myelinsheaths, of nerve cells in the brain and spinal cord are damaged. This damage disrupts the ability of parts of the nervous system to communicate, resulting in a wide range of signs and symptoms, including physical, mental, and sometimes psychiatric problems. MS takes several forms, with new symptoms either occurring in isolated attacks (relapsing forms) or building up over time (progressive forms). Between attacks, symptoms may go away completely; however, permanent neurological problems often occur, especially as the disease advances.
About IntelliCell Biosciences IntelliCell is a pioneering regenerative medicine company focused on the expanding regenerative medical markets using adult autologous stromal vascular fraction cells (SVFCs) derived from the blood vessels in the adult adipose tissue. IntelliCell Biosciences has developed its own patented technology and protocol to separate adult autologous vascular cells from adipose tissue without the use of enzymes. IntelliCell will also be seeking to develop technology-licensing agreements with technology developers, universities, and international business entities.
.0041
alert$$$$
IntelliCell Biosciences Cellular Therapy Treatment Yields Positive Results in Multiple Sclerosis PatientsFont
8:57 AM ET 11/12/13 | Marketwire
IntelliCell Biosciences, Inc. (OTCQB: SVFC), a regenerative medicine company utilizing adult autologous vascular cells (SVCs) derived from the blood vessels found in adipose tissue, announced today that it has supplied its cellular product, stromal vascular fraction cells ("SVFC"), to ReGen Medical PC for the successful treatment of a patient with Multiple Sclerosis.
It is important to note that by "successful," the company is declaring that these cells do not cure Multiple Sclerosis, but of the 19 patients with MS that ReGen Medical has treated using IntelliCell's technology, each have gained anywhere from good to significant quality of life improvements.
In almost every case that ReGen treated, as well as in this case, previous medical history stated the patients' Multiple Sclerosis had progressively worsened. In the case of this patient she had undergone seven years of progression since her original diagnosis. This patient was remitted to the care of Dr. Steven Victor of ReGen Medical PC, where the patient received six treatments of ReGen Medical's proprietary SVFC, which includes stem cells, spaced approximately 3-5 months apart. The results of the treatment reflected significant improvements to the patient's quality of living. Prior to any SVFC treatments, the patient experienced a gradual decline, beginning in 2001, of her vision, bladder control, and mobility, particularly of the extremities on her right side. This culminated in her diagnosis of stage IV multiple sclerosis in 2006. Following the first treatment, the patient regained use of her previously immobile right leg, and coupled with more treatments and physical therapy, the patient can now walk. After her second treatment, the patient noticed more strength and agility in her previously clenched right hand. Additionally, with further treatment the patient noted drastically improved vision in her previously debilitated eye and she regained bladder control. With each patient ReGen Medical has treated, the number of treatments has varied between one and six, each patient experiencing different improvements at various intervals.
Dr. Steven Victor, CEO of IntelliCell Biosciences, commented, "The patient mentioned in this release, was the first clinical case we treated for MS with IntelliCell's SVFC technology. If not for our beneficial treatments, this patient would be restricted to a wheelchair and lose total sight in one of her eyes. Previously, this patient had been unsuccessfully treated at some of the most respected Medical Centers in the country and received little or no improvement to her symptoms. By utilizing IntelliCell's paradigm shifting medical solutions, we have dramatically improved the quality of this patient's life. IntelliCell is confident that the future of medicine will entail utilizing cellular therapy for previously unmet clinical needs, and we expect to establish ourselves as a market leader in this emerging industry."
Multiple Sclerosis (MS), also known as disseminated sclerosis or encephalomyelitis disseminata, is an inflammatory disease in which the insulating covers, called myelinsheaths, of nerve cells in the brain and spinal cord are damaged. This damage disrupts the ability of parts of the nervous system to communicate, resulting in a wide range of signs and symptoms, including physical, mental, and sometimes psychiatric problems. MS takes several forms, with new symptoms either occurring in isolated attacks (relapsing forms) or building up over time (progressive forms). Between attacks, symptoms may go away completely; however, permanent neurological problems often occur, especially as the disease advances.
About IntelliCell Biosciences IntelliCell is a pioneering regenerative medicine company focused on the expanding regenerative medical markets using adult autologous stromal vascular fraction cells (SVFCs) derived from the blood vessels in the adult adipose tissue. IntelliCell Biosciences has developed its own patented technology and protocol to separate adult autologous vascular cells from adipose tissue without the use of enzymes. IntelliCell will also be seeking to develop technology-licensing agreements with technology developers, universities, and international business entities.