visal
In cul@ al nasdaq
- Registrato
- 20/6/01
- Messaggi
- 23.858
- Punti reazioni
- 304
ImmunoCellular Therapeutics Ltd (NYSEMKT:IMUC) extended declines as the Phase II clinical trial evaluation of its ICT-107 vaccine failed to show a statistically significant benefit in the primary endpoint of overall survival, or OS. ICT-107, IMUC’s dendritic cell-based vaccine, did show a statistically significant benefit in progression-free survival (PFS), providing the company support for continuing clinical study of the vaccine in Phase III.
However, MLV & Co. analyst Vernon T. Bernardino believes ICT-107′s approval is likely delayed by at least two years. Therefore, the firm moved its estimate for its approval from 2017 to 2019, and with uncertainty in ICT-107 increased, they seek positive Phase II results with vaccine ICT-140, in ovarian cancer, before becoming buyers of the stock.
MLV & Co. has lowered its rating on the stock from Buy to Hold, and reduced the price target from $6.00 to $1.25.

Se arrivasse a 1.25$ ci metterei la firma



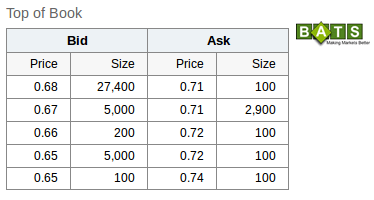

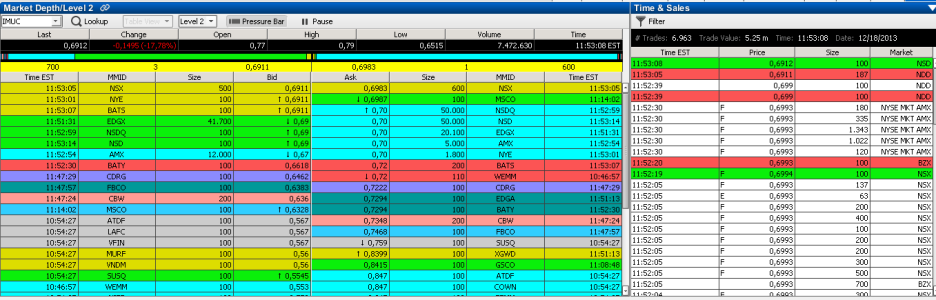

 professionale....
professionale....

