Installa l'app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Nota: This feature may not be available in some browsers.
Stai usando un browser molto obsoleto. Puoi incorrere in problemi di visualizzazione di questo e altri siti oltre che in problemi di sicurezza. .
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
Dovresti aggiornarlo oppure usare usarne uno alternativo, moderno e sicuro.
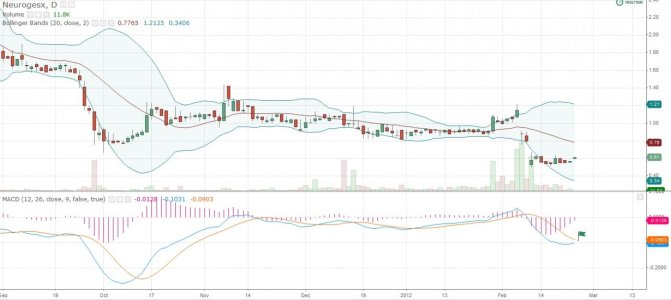
NGSX - NeurogesX, Inc (NASDAQ:NGSX)
- Creatore Discussione coffeman
- Data di inizio
Più opzioni
Chi ha risposto?il supervisore
Nuovo Utente
- Registrato
- 27/11/11
- Messaggi
- 1.565
- Punti reazioni
- 218
questa secondo me è tipo bionovo bisogna saper lucrarci e abbandonarla in tempo
la tentazione di un cippetto è forte ma mi ero promesso di lasciare stare ste porcherie di azien de
la tentazione di un cippetto è forte ma mi ero promesso di lasciare stare ste porcherie di azien de
il supervisore
Nuovo Utente
- Registrato
- 27/11/11
- Messaggi
- 1.565
- Punti reazioni
- 218
okkio ch eparte.. buona per ingresso speculativo
l 'altro giorno ho ceduto alla tentazione e l ho presa a 0,56 devo recuperare un piccolo loss avuto con bionovo
il supervisore
Nuovo Utente
- Registrato
- 27/11/11
- Messaggi
- 1.565
- Punti reazioni
- 218
lovis
Nuovo Utente
- Registrato
- 14/4/01
- Messaggi
- 9.351
- Punti reazioni
- 376
NeurogesX, Inc. (Nasdaq:NGSX) announced that the FDA has accepted for review the its supplemental new drug application (sNDA) for Qutenza (capsaicin) 8 percent, for the management of neuropathic pain associated with HIV-associated peripheral neuropathy (HIV-PN). The FDA has granted Qutenza a priority six month review classification, assigning a Prescription Drug User Fee Act (PDUFA) action date of March 7, 2012.
NGSX ApprovazioneTradingNoStop | TradingNoStop
e domani earning
NGSX ApprovazioneTradingNoStop | TradingNoStop
e domani earning
ivantopastro
Nuovo Utente
- Registrato
- 3/3/08
- Messaggi
- 2.655
- Punti reazioni
- 41
NeurogesX, Inc. (Nasdaq:NGSX) announced that the FDA has accepted for review the its supplemental new drug application (sNDA) for Qutenza (capsaicin) 8 percent, for the management of neuropathic pain associated with HIV-associated peripheral neuropathy (HIV-PN). The FDA has granted Qutenza a priority six month review classification, assigning a Prescription Drug User Fee Act (PDUFA) action date of March 7, 2012.
NGSX ApprovazioneTradingNoStop | TradingNoStop
quando diranno se approvato?
il supervisore
Nuovo Utente
- Registrato
- 27/11/11
- Messaggi
- 1.565
- Punti reazioni
- 218
E questione di tempo 0.9 è il mio target prese tempo addietro a 0.56
serginator
Nuovo Utente
- Registrato
- 30/3/08
- Messaggi
- 94
- Punti reazioni
- 3
scusate, ma oggi non c'è la decisione della fda dell'approvazione? non riesco a trovare la notizia.